Product Images Trifluoperazine Hydrochloride
View Photos of Packaging, Labels & Appearance
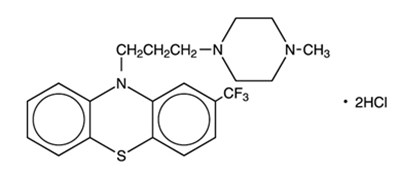
- Structural Formula - 9e1389f4 c477 458f b7de f0f6e3107fe8 01
- Unit Carton - image 01
- Trifluoperazine HCl 2 mg Tablets Unit Carton Label - image 02
- Unit Carton - image 03
- Trifluoperazine HCl 5 mg Tablets Unit Carton Label - image 04
- Unit Carton - image 05
- Trifluoperazine HCl 10 mg Tablets Unit Carton Label - image 06
- Unit Carton - image 07
- Trifluoperazine HCl 1 mg Tablets Unit Carton Label - image 08
Product Label Images
The following 9 images provide visual information about the product associated with Trifluoperazine Hydrochloride NDC 51079-573 by Mylan Institutional Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Unit Carton - image 01

This is a brief description of Trifluoperazine Hydrochloride Tablets, a prescription medication used to treat severe mental disorders. The National Drug Code is 51079-572-20. The tablets come in a blist-pack with one tablet per blister. Each table contains the equivalent of 1 mg of Trifluoperazine Hydrochloride USP. The usual dosage is between 2 and 4 mg per day. Dosages above 4 mg daily are generally not recommended for long-term use. This medication should be stored between 68° and 77°F, protected from light. The manufacturer is Mylan Pharmaceuticals located at Morgantown, United States.*
Trifluoperazine HCl 2 mg Tablets Unit Carton Label - image 02

This is a warning label found on a drug container. The label warns that the unit dose package is not child-resistant and is only available for institutional use. The drugs contained in the package must be kept out of reach of children. The container provides light-resistance and comes from Mylan in Rockford, USA. The lot number and expiration date can be found on the container's window.*
Unit Carton - image 03

This is a description of a medication called Trifluoperazine Hydrochloride. It comes in tablet form and is identified by the codes IDC 51079-57 and NDC 51079-574-20. Each tablet contains 5 grams of Trifluoperazine Hydrochloride USP and is typically taken in doses ranging from 2 to 4 mg daily. The accompanying prescribing information should be consulted for more details. The tablets should be stored between a temperature range of 20°C to 25°C. The remaining text is not readable.*
Trifluoperazine HCl 5 mg Tablets Unit Carton Label - image 04

This is a packaging for a drug produced by Mylan. It is not child-resistant and intended for institutional use only. The container provides light-resistance, and instructions are given to keep it and all drugs out of the reach of children. The lot number and expiration date can be found in a window. No specific information is available regarding the drug itself.*
Unit Carton - image 05

Trifluoperazine Hydrochloride is a medication available in tablet form under the NDC code 51079-573-20. It is used for the treatment of various conditions and diseases, with a usual recommended dose of 2-4mg per day. However, doses greater than this should only be used in severe conditions, as per accompanying prescribing information. Each tablet in this bottle contains 102mg of Trifluoperazine Hydrochloride. Mylan Pharmaceuticals Inc. is the manufacturer of this medication, and the tablets should be stored at controlled room temperature and protected from light.*
Trifluoperazine HCl 10 mg Tablets Unit Carton Label - image 06

This is a package label for a medication from Mylan, Inc. The package is not child-resistant and is intended for institutional use only. It is important to keep all medications out of the reach of children. The container provides light-resistance, which helps preserve the medication. Consumers can check the lot number and expiration date by looking in the designated window on the package.*
Unit Carton - image 07

This is a description for a medication called "Tablets, USP" which is only available by prescription. Each bottle contains 100 tablets (10x10) that are coated and contain Tioperazne and iectiode (amount unspecified) with a maximum daily dosage of 4mg. The medication must be protected from light and stored at controlled room temperature. No additional information is available due to poor quality.*
Trifluoperazine HCl 1 mg Tablets Unit Carton Label - image 08

This is a package of medicine meant for institutional use only. It should be kept away from the reach of children because it is not child-resistant. The container is designed to provide resistance to light. The lot number and expiration date can be found on the window of the container. The medication is manufactured by Mylan located in Rockford, USA. There is no information available about what the medicine is for as it is not mentioned in the text.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.