Product Images Buspirone Hydrochloride
View Photos of Packaging, Labels & Appearance
- Structural Formula - 103f116c e4f4 47d7 82aa 59a2962e465e 01
- 15 mg (the entire tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 02
- 10 mg (two-thirds of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 03
- 5 mg (one-third of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 04
- 7.5 mg (one-half of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 05
- 30 mg (the entire tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 06
- 20 mg (two-thirds of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 07
- 10 mg (one-third of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 08
- 15 mg (one-half of a tablet) - 103f116c e4f4 47d7 82aa 59a2962e465e 09
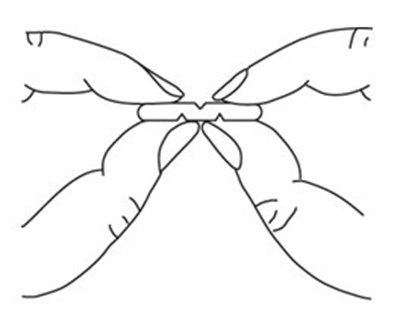
- Breaking Multi-Scored Tablet - image 01
- Buspirone Hydrochloride 5 mg Tablets Unit Carton Label - image 02
- Unit Carton - image 03
- Buspirone Hydrochloride 10 mg Tablets Unit Carton Label - image 04
- Unit Carton - image 05
- Buspirone Hydrochloride 15 mg Tablets Unit Carton Label - image 06
- Unit Carton - image 07
- Buspirone Hydrochloride 30 mg Tablets Unit Carton Label - image 08
- Unit Carton - image 09
- image 10
Product Label Images
The following 19 images provide visual information about the product associated with Buspirone Hydrochloride NDC 51079-960 by Mylan Institutional Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Buspirone Hydrochloride 5 mg Tablets Unit Carton Label - image 02

This is a description of a medication called busPIRone Hydrochloride Tablets, USP. The National Drug Code (NDC) of the medication is 51079-985-20. It is Rx only and comes with 100 tablets per bottle. Each tablet contains 5mg of Buspirone hydrochloride, USP. The text includes instructions that usual dosage should be taken as per accompanying prescribing information. The medication must be stored at a temperature between 20° to 25°C (68° to 77°F) and protected from light. The medication is manufactured by Mylan Pharmaceuticals Inc. located in Morgantown, Wy 26505 US A.*
Unit Carton - image 03

This text describes a medication called busPIRone Hydrochloride that comes in tablet form. The tablets are labeled with the NDC code 51079-986-20 and come in a bottle of 100 tablets. Each tablet contains 10mg of Buspifone hydrochloride. There are instructions on the label on how to take the medication and how to store it. The medication is manufactured by Mylan Pharmaceuticals Inc. in the United States.*
Buspirone Hydrochloride 10 mg Tablets Unit Carton Label - image 04

This unit dose package is not child resistant. It is intended for institutional use only. Please keep this and all drugs out of the reach of children. This container provides light resistance. To see the lot number and expiration date, refer to the window. Mylan® S v is located in Rockford, IL 61103 USA.*
Unit Carton - image 05

busPIRone Hydrochloride Tablets, USP, containing 15mg of Buspirone hydrochloride per tablet. It is advised to refer to the prescribing information and patient instruction sheet for the usual dosage. They should be stored at a temperature of 20° to 25°C, protected from light. Manufactured by Mylan Pharmaceuticals Inc. in Morgantown, USA.*
Buspirone Hydrochloride 15 mg Tablets Unit Carton Label - image 06

NDC 51079-994-20 are tablets containing Buspirone hydrochloride, used as prescription medicine. Each bottle contains 100 tablets and the recommended dosage can be found in the prescribing information and patient instruction sheet. The tablets should be stored at controlled room temperature and protected from light. The manufacturer is Mylan, located in Morgantown, W 26505 USA.*
Unit Carton - image 07

This text is a warning label regarding medication packaging. It notes that the unit dose package is not child-resistant and should be kept out of reach of children. Additionally, it advises that the container provides light-resistance and includes a window with the lot number and expiration date. The medication is manufactured by Mylan and the address for their facility is listed at the end.*
Buspirone Hydrochloride 30 mg Tablets Unit Carton Label - image 08

This is a medication package that is not child-resistant and is intended only for institutional use. It is important to keep this and all medications out of children's reach. The package provides light-resistance and the number and expiration date can be found on the window. The medication brand is Mylan® S v and the package was made in Rockford, L 61103 USA.*
Unit Carton - image 09

This is a notice regarding a non-child resistant unit dose package intended for institutional use only. There is a caution to keep all drugs, including this package, out of the reach of children. The container provides light-resistance and there is a window displaying the lot number and expiration date. The product is called Mylan S and has been manufactured in Rockford, Illinois, USA.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.