Product Images Ultracare Oral Anesthetic Bubble Gum
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Ultracare Oral Anesthetic Bubble Gum NDC 51206-201 by Ultradent Products, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure - ultracare 03
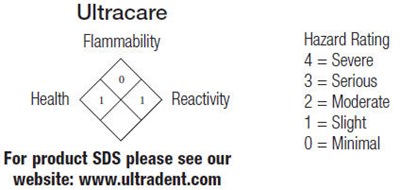
The text is a description for Ultracare, a product with information about its flammability hazard rating, health, and reactivity. It also provides a reference to the company's website for additional information in SDS format. The numbers 0 indicate a minimal hazard.*
Key - ultracare 04
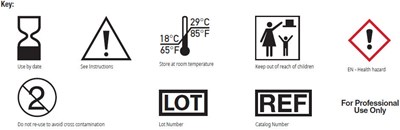
This text is a mix of various warnings and instructions related to a unidentified product. It advises to store the product at room temperature and mentions an expiration date that should be checked in the instructions. It also warns that the product may represent a health hazard and should be kept out of reach of children. In addition, it suggests not to reuse the product to avoid cross-contamination and identifies the product with a lot reference and catalog number.*
PRINCIPAL DISPLAY PANEL - 30 mL Syringe Label - ultracare 05

Drug Facts for Bonzocaing 20% wh. It is used as a temporary relief for oral pain caused by orthodontic appliances. There are warnings about potential allergic reactions to the drug, sore throat, and to seek medical help if necessary. Some of the text is not decipherable.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

