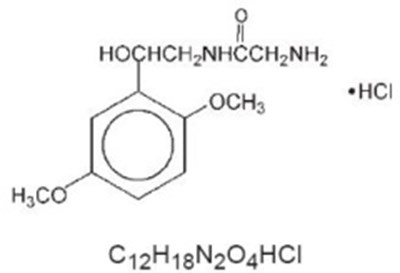
Product Images Midodrine Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Midodrine Hydrochloride NDC 51407-388 by Golden State Medical Supply, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
51407-388-90LB.jpg - 51407 388 90LB

This is a description for a medication identified as Midodrine Hydrochloride Tablets, USP. It contains 2.5mg and has 5 tablets. There are dosing information and tamper temperature instructions included in the package insert. The manufacturer is GSMS Incorporated, with the NDC number 51407-388-30.*
51407-389-90LB.jpg - 51407 389 90LB

This is a product description for Midodrine Hydrochloride, a medication that comes in the form of tablets. The tablets contain 5mg of the active ingredient and are sold in packages of 90 units. The package insert contains dosage information. The product is manufactured by GSMS Incorporated and has a control temperature range of 20°C to 25°C or 68°F to 77°F.*
51407-390-90LB.jpg - 51407 390 90LB

This appears to be a mix of text fragments and incomplete words, likely from a product label or insert. The only clear information that can be extracted is the name "GSMS Incorporated" and the drug name "Midodrine Hydrochloride" in tablet form. The NDC code is also partially visible as "NDC51407-390-90". Additional dosing instructions are likely included in the package insert, which is referenced at the end. Without proper formatting and complete sentences it is difficult to provide a more detailed description.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.