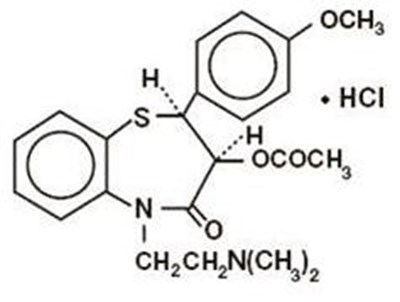
Product Images Diltiazem Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Diltiazem Hydrochloride NDC 51407-474 by Golden State Medical Supply, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
51407-473-90OL.jpg - 51407 473 90OL

This is a medication called Diltiazem Hydrochloride that comes in the form of extended-release capsules with a strength of 120mg. It should be kept in a tightly sealed container away from excessive humidity and children. The capsule should not be accepted if the seal over the bottle opening is broken or missing. The recommended dosage can be found in the accompanying Prescribing Information or per Once-a-Day Dosage. It is manufactured by TWi Pharmaceuticals, Inc. and marketed by GSMS, Incorporated in the USA. The label includes the important information needed to use the medicine safely.*
51407-474-90OL.jpg - 51407 474 90OL

This is a medication description for dilTIAZem Hydrochloride Extended-Release Capsules, USP, containing 180mg per capsule, and is taken once daily. It comes in a package of 90 capsules and should be stored between 68°-77°F in a tightly sealed, light-resistant container. The medication should be kept out of reach of children and away from excessive humidity. This medication is manufactured by TWi Pharmaceuticals, Inc. and marketed by GSMS, Incorporated. For more detailed information on dosages, refer to the accompanying Prescribing Information.*
51407-475-90OL.jpg - 51407 475 90OL

NDC 51 407-475-90 is an Extended-Release capsule containing 240mg of Diltiazem Hydrochloride. The recommended dosage must be obtained from the accompanying prescribing information. The capsule is light-resistant and should be kept in a controlled room temperature. The medication should be kept out of reach of children. The container should not be accepted if the seal over the bottle opening is broken or missing. The medication is manufactured by TWi Pharmaceuticals and marketed by GSMS, Incorporated.*
51407-476-90OL.jpg - 51407 476 90OL

This is a medication with NDC 51 407-476-90 that comes in the form of capsules. Each capsule contains 300mg of Diltiazem Hydrochloride in an extended-release formulation. The medication should be dispensed in a light-resistant container and should be kept away from excessive humidity. The medication should not be used if the seal over the bottle is broken or missing. This medication is manufactured by TWi Pharmaceuticals, Inc. and marketed by GSMS, Incorporated. The recommended dosage for this medication is once-a-day. Store in a controlled room temperature between 20°C to 25°C (68°F to 77°F).*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.