Product Images Lurasidone Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Lurasidone Hydrochloride NDC 51407-836 by Golden State Medical Supply, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
51407-836-05LB - Lurasidone HCI - Rev. 0324.jpg - 51407 836 05LB Lurasidone HCI Rev. 0324

This is information regarding a medication called Lurasidone Hydrochloride in tablet form. Each tablet contains 20 mg of lurasidone HCI. It includes instructions for pharmacists to dispense a Medication Guide to each patient and store the medication in a tight, light-resistant container. The medication should be kept out of the reach of children and stored at 20° - 25°C (68° - 77°F). The contact information for the marketer is GSMS, Incorporated Camarillo, CA 93012 USA.*
51407-837-05LB - Lurasidone HCI - Rev. 0324.jpg - 51407 837 05LB Lurasidone HCI Rev. 0324

The text provides information on Lurasidone Hydrochloride tablets with 60 mg strength, each tablet equivalent to 55.87 mg of lurasidone. The medication guide should be dispensed to each patient, and the complete prescribing information should be followed for dosage. The tablets are to be stored in a tightly closed, light-resistant container at room temperature. The medication is available in a bottle containing 500 tablets and is for prescription use only. Further information and the medication guide can be found at the provided URL.*
51407-838-05LB - Lurasidone HCI - Rev. 0324.jpg - 51407 838 05LB Lurasidone HCI Rev. 0324

Each tablet contains 120 mg of lurasidone HCI (equivalent to 111.73 mg of lurasidone). This medication is dispensed in a tight, light-resistant container as per USP standards. It is important to keep the container tightly closed and out of reach of children. The pharmacist should provide a Medication Guide to each patient and store the medication at a temperature between 20°C to 25°C (68°F to 77°F). For more information, consult the accompanying Prescribing Information.*
figure-1 - figure 1
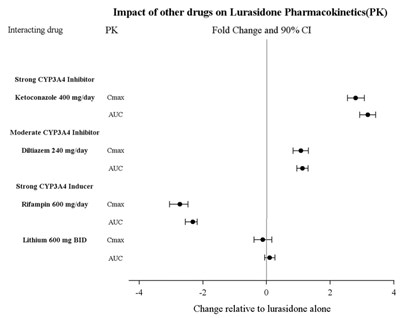
The text mentions the impact of different drugs on the pharmacokinetics of Lurasidone. It provides information on the fold change and 90% confidence intervals for drugs categorized as Strong CYP3A4 Inhibitors (Ketoconazole), Moderate CYP3A4 Inhibitors (Diltiazem), and Strong CYP3A4 Inducers (Rifampin). The text also references the effect of Lithium on Cmax and AUC. The data indicates how these drugs influence Lurasidone pharmacokinetics (PK) relative to Lurasidone alone.*
figure-2 - figure 2
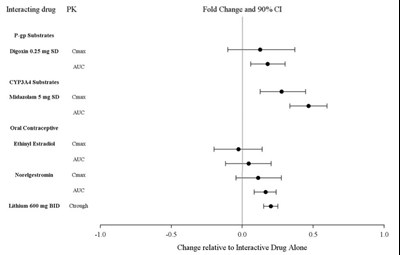
This text provides information on interacting drugs, including P-gp substrates like digoxin, CYP3A4 substrates such as midazolam, an oral contraceptive containing ethinyl estradiol and norelgestromin, and lithium. It also mentions pharmacokinetic parameters like Cmax, AUC, and Ctrough, along with fold change and 90% confidence interval. The data indicates changes in these parameters when these drugs are taken together.*
figure-3 - figure 3
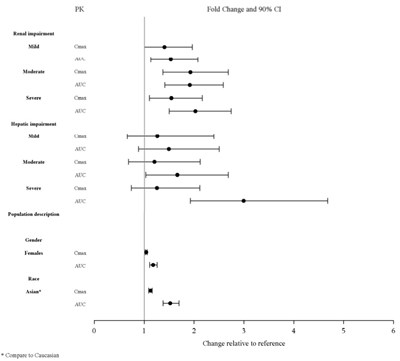
This text seems to contain information about renal impairment, gender comparison between Asian and Caucasian populations in relation to pharmacokinetics, and variability in fold change relative to reference values. It provides insights into the impact of renal and hepatic impairment on moderate to severe levels and their association with pharmacokinetics. Additionally, it suggests the comparison of these factors between different ethnic groups.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.