Product Images Lovastatin
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Lovastatin NDC 51655-686 by Northwind Pharmaceuticals, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Label - 51655 686 52 Master Bottle Label Approval Rev A 04 21

Lovastatin is a tablet used for medical purposes, each containing 10mg of the active ingredient. The tablets come in a bottle of 30 and require a prescription. They should be kept in their original container, protected from light and stored at a controlled room temperature of 20°-25°C (68-77°F). Lovastatin is repackaged by the Northwind Pharmaceuticals in Indianapolis and sourced from Carlsbad Technology in lot number 0000000000. The GTIN number is 0035165686522 and the S/N is 000000000000000. The expiration date is 00/00/0000.*
Figure 1 - lov00 0004 03
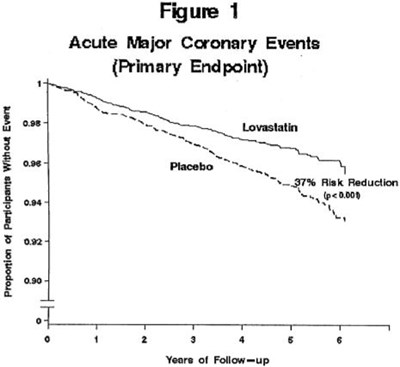
The text describes a figure (Figure 1) related to the proportion of participants without an acute major coronary event over a follow-up period of 1 to 4 years. It appears to be related to a primary endpoint of a study, but no further information is available.*
Table VII - lov00 0005 04
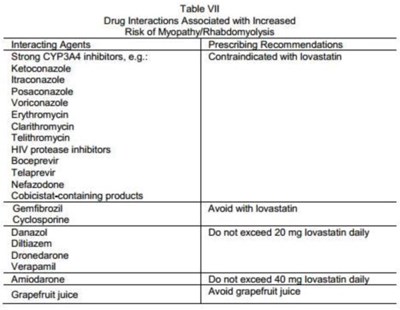
The given text provides a table of drug interactions that are associated with increased risk of myopathy/rhabdomyolysis. The table lists several prescribing recommendations for various drugs such as strong CYP3A4 inhibitors, HIV protease inhibitors, Gemfibrozil, Cyclosporine, Danazol, Difiazem, Dronedarone, and grapefruit juice. The recommended doses and usage for each drug are also mentioned in the table.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

