Product Images Adenosine Injection, 6mg/2ml (3mg/ml) Vial
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Adenosine Injection, 6mg/2ml (3mg/ml) Vial NDC 51662-1201 by Hf Acquisition Co. Llc, Dba Health First, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Adenosine Flag Label - 20180711 hf adenosine glint flag label 1 July 11 Update resized

This appears to be a label or packaging for a medical device or medication. It includes a product code (10), a location (Mukilteo, WA), and a warning that the product is only available by prescription (RX ONLY). However, without additional information or context, it is not clear what specific product this label is attached to.*
5166212012 label

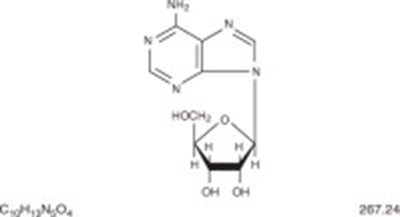
Adenosine Injection is a solution for rapid bolus intravenous use that comes in a 6mg/2mL (3mg/mL) single-use vial. This medication does not contain any preservatives and any unused portion after opening should be discarded. It is essential to keep the injection away from children and at a specific storage temperature range of 20 to 25C (68 to 77F) to avoid crystalization. However, if the solution crystallizes, it can be dissolved by warming to room temperature. The manufacturer's instruction should be followed closely, and the solution must be clear and particle-free while in use. The Lot and Expiration date for this particular product are ABCDEFGHIJKLMNOPQRST and 2021-06-01, respectively. The National Drug Code (NDC) for this injection is 51662-1201-2, and the Original Manufacturer's NDC is 17478-542-02. This medication is available only by prescription, and HF Acquisition Co., LLC distributes it.*
5166212013 label

Adenosine Injection, USP is a solution used for rapid intravenous administration without any preservatives. The solution should be kept out of reach of children and should be discarded if unused. It should be stored at room temperature (20 to 25°C) and must not be refrigerated as it can cause crystalization. In case of crystalization, the solution should be warmed to room temperature until the crystals are dissolved. The solution must be clear and free from particles at the time of use. The manufacturer’s package insert should be referred to for detailed information. The product is distributed by HF Acquisition Co., LLC, and has the NDC code 51662-1201-3. The lot number is ABCDEFGHIJKLMNOPGQRST and the expiry date is 2021-06-01.*
Anenosine Container Label - Adenosine Container Label

This is a description of a medication called Adenosine. It is an injection that is used intravenously for rapid bolus treatment. The text contains various alphanumeric codes, but no substantial information about side effects or indications for use.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.