Product Images Epinephrine
View Photos of Packaging, Labels & Appearance
- 00351662122129 Epinephrine pouch label
- 00351662122136 Epinephrine RFID label
- 00351662122136 Epinephrine case label (1)
- SERIALIZED BOX - 51662 1221 1 Epinephrine Injection 10 Serialized Label
- HOW SUPPLIED LOGO - How Supplied Logo
- HOW SUPPLIED TABEL - How Supplied
- LOGO - Logo
- STRUCTURE - Structure
- CARTON - Updated 0409 4921 20 Epinephrine SYR Box Label
- SYRINGE - Updated Syringe
Product Label Images
The following 10 images provide visual information about the product associated with Epinephrine NDC 51662-1221 by Hf Acquisition Co Llc, Dba Healthfirst, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
00351662122129 Epinephrine pouch label

EPINEPHRINE INJECTION, USP 1mg/10mL is a warning label on a medical prescription that contains sulfites and needs protection from light. The unused portion should be discarded, and the drug should not be administered if its color is pinkish or darker than slightly yellow or if it contains any precipitate. The usual dosage for the medication is provided in the insert. The injection is for intravenous use only, and the temperature for storage is between 20 to 25°C (68 to 77°F). The drug is manufactured by HF Acquisition Co., LLC, located in Mukilteo, WA 98275.*
00351662122136 Epinephrine case label (1)
.jpg)
EPINEPHRINE INJECTION, USP 1mg/10mL is a medication that contains sulfites and should be protected from light. Unused portions should be discarded, and it should not be used if it appears pinkish or darker than slightly yellow or if it contains a precipitate. The usual dosage can be found on the insert, and it should be stored at 20 to 25°C (68 to 77°F). The manufacturer is HF Acquisition Co., LLC, located in Mukilteo, WA, and its original NDC is 0409-4921-34. It is available by prescription only.*
HOW SUPPLIED TABEL - How Supplied
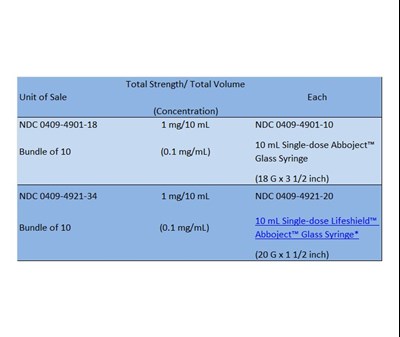
This is a product with a National Drug Code of 0409-4901-18, which contains 1mg of medication per 10mL. There is also a National Non-proprietary name Drug Code of 0409-4901-10 for this medication. The product is a bundle of 10 single-dose Abboject™ syringes, each containing 0.1mg/mL of medication. The syringes are made of glass and have a needle size of 18 G x 3 1/2 inch.*
SYRINGE - Updated Syringe

This is a description of a medical product, which is a 1 mg/10 mL Epinephrine injection, sterile and non-pyrogenic, with a pH of 3.3 (2.2 to 5.0). The product is intended for intravenous use and comes in single-dose vials of 210 mL, identified by the National Drug Code (NDC) number 0409-4921-20. The label indicates that the product is available only by prescription (Rxonly) and includes Hospira, Inc. as the manufacturer with their contact address in Lake Forest, IL 60045, USA. The text also mentions that the package includes an insert providing dosage and administration instructions.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.





