Product Images Naloxone Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Naloxone Hydrochloride NDC 51662-1238 by Hf Acquisition Co. Llc, Dba Healthfirst, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
CARTRIDGE - 1000420 0409 1782 03 Naloxone Carp
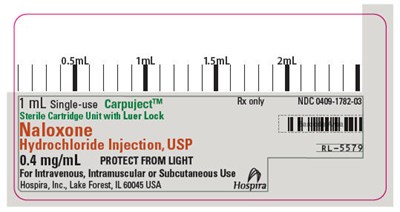
This is a description of a single-use cartridge unit with 0.4 mg/mL of Naloxone Hydrochloride Injection, USP, labeled with the code "1M TV T". It is intended for sterile use with intravenous, intramuscular or subcutaneous administration, and has a Luer Lock. The manufacturer of this product is Hospira, Inc., located in Lake Forest, Illinois, USA.*
SERIALIZED LABEL - 51662 1238 1 Naloxone HCL Injection

This is a description of a medicine named Naloxone, which is distributed by HF Acquisition Co. The text contains some identification numbers and codes of the product, and recommends the user to see the manufacturer's package insert. The product is an injection with a concentration of 0.4 mg/mL and can be used for protection.*
HOW SUPPLIED - How Supplied

This appears to be a description of a product for sale. The product is a box of cartridges, each containing either 1mL or 2.5mL of a solution at 0.4mg/mL. The product is designed to be used with the Carpuject Syringe System and comes with a Luer Lock. The price is not mentioned.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

