Product Images Epinephrine
View Photos of Packaging, Labels & Appearance
- 51662-1320-2 Package Labeling - 51662 1320 2 Exterior Package Labeling
- 51662-1321-2 Product Package Labeling - 51662 1321 2 Product Package Labeling
- Serialized Product Labeling - 51662 1321 2 Serialized Product Labeling
- FIGURE A - A
- AUVI-Q 0.15MG CASE - AUVI Q 1321 LABEL 1
- AUVI-Q 0.15mg DEVICE LABEL - AUVI Q 1321 LABEL 2
- AUVI-Q CASE LABEL - AUVI Q LABEL 1
- AUVI-Q DEVICE LABEL - AUVI Q LABEL 2
- FIGURE B - B
- FIGURE C - C
- FIGURE D - D
- FIGURE E - E
- SERIALIZED LABEL - SERIALIZED 51662 1320 1 Auvi Q 0.3mg
- SERIALIZED 0.15MG - SERIALIZED
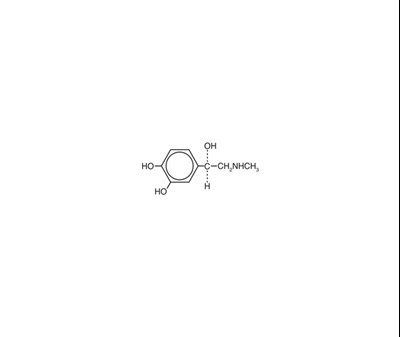
- STRUCTURE - STRUCTURE
- Serialized 51662-1320-2 - Serialized Product Labeling 51662 1320 2
- TRAINER INST. - TRAINER INST
- Trainer Part 1 - Trainer Part 1
- Trainer Part 2 - Trainer Part 2
Product Label Images
The following 19 images provide visual information about the product associated with Epinephrine NDC 51662-1321 by Hf Acquisition Co Llc, Dba Healthfirst, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
51662-1320-2 Package Labeling - 51662 1320 2 Exterior Package Labeling
This is a product description for a single-use epinephrine injection device called Auvi-Q, used for patients experiencing allergic emergencies. It contains instructions for use and information on dosage and storage. The device should not be refrigerated or frozen and must be replaced if the solution is discolored or cloudy. Information on the manufacturer and the ingredients is also included.*
51662-1321-2 Product Package Labeling - 51662 1321 2 Product Package Labeling
Auvi-Q Trainer is a device designed for single-use injection, used for allergic emergencies. The kit includes a Patient Information leaflet, Trainer Instructions for use insert, and does not contain an active drug or needle. The epinephrine injection in the kit, containing 0.15mL, contains 0.5mg epinephrine, 1.2mg sodium chloride, and is manufactured by kaléo, Richmond, VA, USA. The device does not require refrigeration and should be protected from heat and light. If the solution is discolored, cloudy, or contains particles, the auto-injector should be replaced.*
FIGURE A - A

This text is a scattered unformatted set of information, but we can assume that it might be related to a physical device. The device's name could be "Device Quter Case". The device seems to have a viewing window, an LED, and a red safety guard that surrounds a needle end that sits on a black base. The information seems incomplete and difficult to draw any further conclusions from it.*
AUVI-Q 0.15MG CASE - AUVI Q 1321 LABEL 1
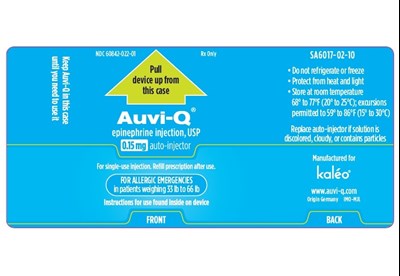
This is a description for a medication called "epinephrine injection, USP" indicated for allergic emergencies in patients weighing between 33 to 66 pounds. The NDC number is 60842-022-01, and it comes in a single-use auto-injector. The medication should not be refrigerated or frozen and should be stored at room temperature between 68° to 77°F. The instructions for use are found inside the device. The medication should be refilled with a prescription after use. It is manufactured for kaléo and the website is www.auvi-g.com. The text also includes a lot number and an expiration date.*
AUVI-Q 0.15mg DEVICE LABEL - AUVI Q 1321 LABEL 2
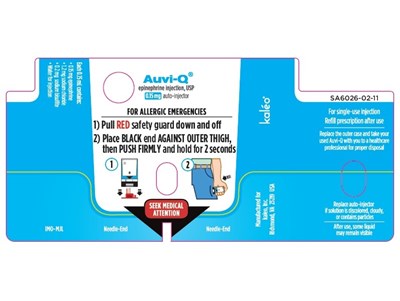
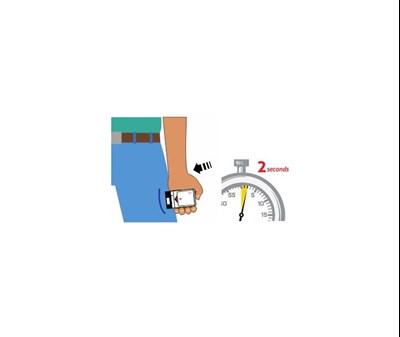
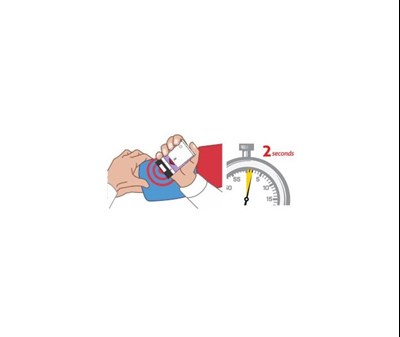
This is a description of Auvi-Q°, an auto-injector used in allergic emergencies. The instructions state to pull the red safety guard down, place the black end against the outer thigh, push firmly, and hold for 2 seconds. Medical attention should be sought after use, and the device is for single-use only. The outer case should be replaced, and the device should be taken to a healthcare professional for proper disposal or if any fluid remains visible.*
AUVI-Q CASE LABEL - AUVI Q LABEL 1
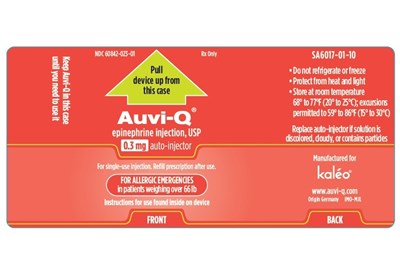
Description: This is a medication product description for epinephrine injection, USP. It includes details such as the NDC code, storage and usage instructions, dosage, and the manufacturer's name. It is meant for emergency use for allergic reactions in patients weighing over 66lb. The instructions for use are found inside the device. It is not recommended to refrigerate or freeze the product, and it should be protected from heat and light. The product should also be replaced if the solution is discolored, cloudy, or contains particulates.*
AUVI-Q DEVICE LABEL - AUVI Q LABEL 2
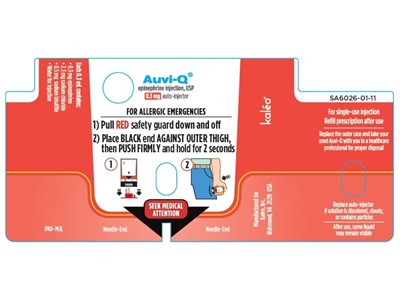
Auvi-Q is an epinephrine injection USP with an auto-njector to be used in case of allergic emergencies. The injection comes with a red safety guard that needs to be pulled down and off before using it. The black end of the auto-njector should be placed against the outer thigh, pushed firmly, and held for two seconds. Seeking medical attention is recommended afterwards. The also provided an address that could be potentially related to the product: Richmond, VA 379 Ush.*
SERIALIZED LABEL - SERIALIZED 51662 1320 1 Auvi Q 0.3mg

Auvi-Q is a USP epinephrine injection auto-injector, used for single-use injection in patients weighing over 66 Ib in allergic emergencies. This device is distributed by HF Acquisition Co., LLC of Mukilteo, WA. Its NDC number is 60842-023-01, and other identifying text includes the codes (01)00351662132012, (17)191030, (21)95410914137, and (10)000005116198. Instructions for use are provided inside the device, and the manufacturer's package insert should be consulted. The user should refill the prescription after use.*
Serialized 51662-1320-2 - Serialized Product Labeling 51662 1320 2

This is a description of an epinephrine auto-injector used for allergic emergencies. It contains 0.3mg of epinephrine injection, USP and is pre-filled. The product might contain batteries with special handling requirements.*
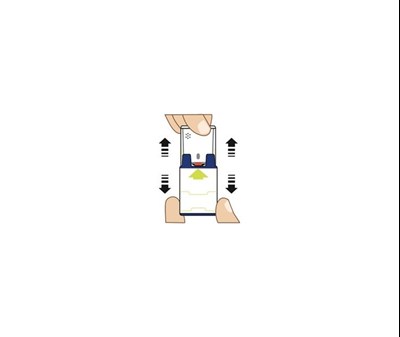
TRAINER INST. - TRAINER INST
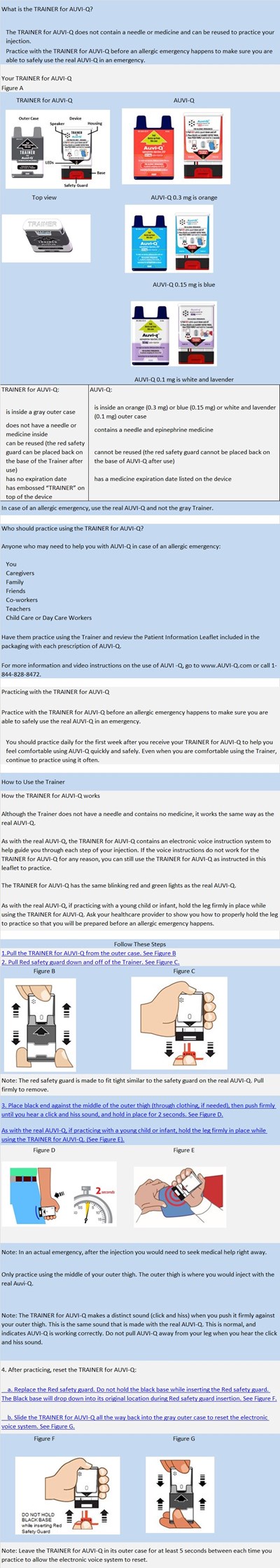
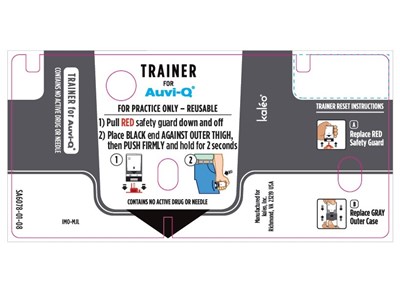
The TRAINER for AUVI-Q is a device used to practice the safe injection of AUVI-Q in case of an allergic emergency. It does not contain a needle or medicine and can be reused. Anyone who may need to help in an emergency, such as caregivers, family members, co-workers, teachers, or child care workers, can practice using the Trainer. The device has electronic voice instructions and blinking red and green lights, and it makes a distinct sound (click and hiss) when used correctly. After practicing, the device must be reset by replacing the red safety guard and sliding it all the way back into the gray outer case to reset the electronic voice system. The TRAINER should be used daily for the first week after receiving it and often afterwards to ensure familiarity in case of an emergency.*
Trainer Part 1 - Trainer Part 1
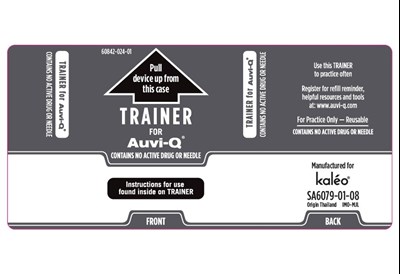
This appears to be a product label or manual for a training device called "TRAINER for Auvi-Q". The device seems to be used for practicing a medical injection procedure and it does not contain any active drug or needle. The label also provides information on where the device is manufactured and how to register for refills and helpful resources.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.