Product Images Lidocaine Hcl And Epinephrine
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 14 images provide visual information about the product associated with Lidocaine Hcl And Epinephrine NDC 51662-1504 by Hf Acquisition Co Llc, Dba Healthfirst, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
51662-1504 50 mL SERIALIZED VIAL LABELING - 51662 1504 1 Serialized Labeling

This is a description of a medical product -- an injection solution containing HCl 2% and Epinephrine 1:100,000, intended for infiltration and nerve block use only, not for epidural or caudal use. It is distributed by HF Acquisition L0, LEC in Mukilteo, WA 93275, and the manufacturer's package insert should be consulted. It is a USP solution and can only be obtained via RX. The LOT number is 09-453-EV and the expiration is in one year.*
51662-1504 50 mL ALTERNATE VIAL LABEL - 51662 1504 1 Vial Label Alternate

This appears to be a label or prescription information for a solution that contains Lidocaine HCI 2% and Epinephrine 1:100,000. The solution is for injection and appears to be in compliance with USP standards. The rest of the text is not clear, with some characters missing or garbled, so a more detailed description cannot be provided.*
51662-1504 50 mL VIAL LABEL - 51662 1504 1 Vial Label

This is a 50mL injection of Lidocaine HCI 2% and Epinephrine 1:100,000 intended for infiltration and nerve block. It is not for use in epidural or caudal procedures. The product is distributed by Hospira Inc. in Lake Forest, IL and is a multiple-dose prescription item. Each mL contains 20mg of anhydrous Lidocaine HC, 10mcg of epinephrine, 6mg of NaCl, 0.5mg of sodium metabisulfite, 0.2mg of anhydrous citric acid as stabilizers, and 1mg of methylparaben added as a preservative. The pH level ranges from 3.3 to 5.5, contains sterile and nonpyrogenic substances and may be adjusted with NaOH and/or HCI. The usual dosage should be checked in the insert, and it should not be autoclaved.*
51662 1504 2 00351662150429 Lidocaine pouch label

This text is a description of an injection solution containing Lidocaine HCI 2% and Epinephrine 1:100,000, which comes in a 50mL vial that is sterile and nonpyrogenic. The solution is not to be used for infiltration or nerve block via the autoclave method or caudal use. The manufacturer's package insert should be consulted before use. The product is to be used only by prescription. The solution is manufactured by HF Acquisition Co. and the physical address is given as "IEO, WA 98275".*
51662 1504 3 00351662150436 Lidocaine case label

This is a product description for Lidocaine HCI 2% and Epinephrine 1:100,000 injection, USP. It comes in a 50mL vial and is sold in boxes of 25. The product is sterile and nonpyrogenic and is intended for infiltration and nerve procedures. The text also includes lot number and expiration date information. This product can only be obtained with a prescription.*
DESCRIPTION - Description
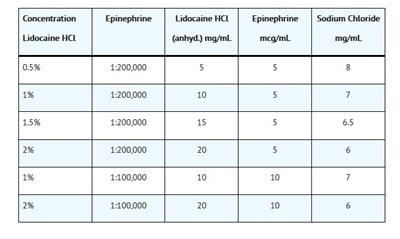
This is a concentration chart showing the amount of Lidocaine HCL, Epinephrine, and Sodium Chloride in different solutions measured in mg/mL. The solutions include different percentages of Lidocaine HCL and varying ratios of Epinephrine concentration. However, without any context, it is not possible to determine how this concentration chart is useful.*
DOSAGE - Dosage

This is a table showing recommended dosages for Lidocaine Hydrochloride injection in various procedures such as infiltration, peripheral nerve blocks, and sympathetic nerve blocks. The recommended dosages are given based on concentration, volume, and total dose in milligrams. The table also provides dosage recommendations for obstetrical analgesia, surgical anesthesia, and analgesia. For dermatomes to be anesthetized, the recommended dose varies from 2 to 3 mL per dermatome.*
HOW SUPPLIED - How Supplied 1

This text appears to be a list of different types of Lidocaine HCL and Epinephrine solutions available for sale. The descriptions include details regarding the concentration and packaging for different unit sizes such as cartons, trays, and cases, each containing varying numbers of fliptop vials or ampuls of the solution. The text also includes a specific product for an Epidural Test Dose in this category.*
PRECAUTIONS - Precautions 1

This is a list of various drugs categorized into different classes, including nitrates/nitrites, local anesthetics, antineoplastic agents, antibiotics, antimalarials, anticonvulsants, and other drugs such as acetaminophen, metoclopramide, quinine, and sulfasalazine. There is no further useful information provided.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




