Product Images Lamotrigine
View Photos of Packaging, Labels & Appearance
- Chemical Structure - lamotrigine 01
- Figure 1 - lamotrigine 02
- Figure 2 - lamotrigine 03
- 25 mg Tablet - lamotrigine 04
- 100 mg Tablet - lamotrigine 05
- 150 mg Tablet - lamotrigine 06
- 200 mg Tablet - lamotrigine 07
- PRINCIPAL DISPLAY PANEL - 25 mg Tablet Bottle Label - lamotrigine 08
- PRINCIPAL DISPLAY PANEL - 100 mg Tablet Bottle Label - lamotrigine 09
- PRINCIPAL DISPLAY PANEL - 150 mg Tablet Bottle Label - lamotrigine 10
- PRINCIPAL DISPLAY PANEL - 200 mg Tablet Bottle Label - lamotrigine 11
Product Label Images
The following 11 images provide visual information about the product associated with Lamotrigine NDC 51672-4133 by Sun Pharmaceutical Industries, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2 - lamotrigine 03

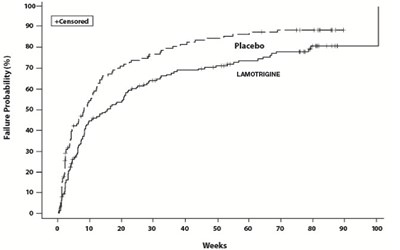
This document seems to be a visual representation of the probability of failure given the use of a placebo versus the use of a medication called "lamotrigine". It is not clear what this failure pertains to, and the rest of the content is not available.*
PRINCIPAL DISPLAY PANEL - 25 mg Tablet Bottle Label - lamotrigine 08

This is a description of a pharmaceutical product called "Lamotrigine". It comes in the form of 100 tablets, each containing 25 mg of the active ingredient "fdtigite". The text includes prescribing information and instructions to store the tablets in a tight container at a temperature between 20°C to 25°C. The product is distributed by Taro Pharmaceuticals USA and caution is advised to verify the product before dispensing.*
PRINCIPAL DISPLAY PANEL - 100 mg Tablet Bottle Label - lamotrigine 09

This product is a tablet of 100 mg of antihistamine, manufactured by Taro Pharmaceuticals. The tablets come in a bottle containing 100 tablets. There are some cautionary notes about verifying the product that has been dispensed as prescription-only medication. The text includes details about Taro Pharmaceuticals including its address in Israel and its US distributor. It also provides storage instructions to protect the tablets from certain temperatures.*
PRINCIPAL DISPLAY PANEL - 150 mg Tablet Bottle Label - lamotrigine 10

This is a medication with NDC code 51672-4132-4, containing 60 tablets of Lamotrigine USP. Each scored tablet has 150 mg of the medication. The package includes prescribing information and a medication guide for the patient. The medication should be stored in a dry place with a temperature between 20°C to 25°C (68°F to 77°F) and protected from light. It should be dispensed in a light-resistant container as defined in the USP. The manufacturer is Taro Pharmaceuticals and the trademark is TARDI.*
PRINCIPAL DISPLAY PANEL - 200 mg Tablet Bottle Label - lamotrigine 11

Each tablet of Ach Scored contains 200 mg of the drug Amigotine, and it is identified by the NDC code 51672-4133-4. The drug should be stored at room temperature in a dry and safe place. The medication guide should be provided to each patient, and it should be dispensed in a container that resists light. The product is distributed by Taro Pharmaceuticals USA Inc. located in Hawthorne, New York. The last line is unclear.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.