Product Images Lazanda
View Photos of Packaging, Labels & Appearance
Product Label Images
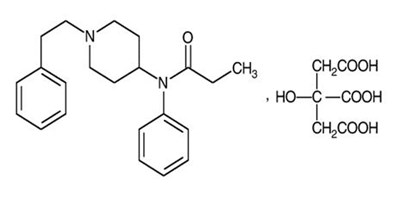
The following 17 images provide visual information about the product associated with Lazanda NDC 51772-311 by Archimedes Pharma Us Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Flow Chart - lazanda 01

This text describes the titration process for Lazanda, a medication for pain relief that is administered through the nostrils. The starting dose is 100 meg and the user is instructed to wait at least two hours before using it again for the next episode of pain. The user is asked to evaluate the pain relief after 30 minutes and based on the response, instructions are given to either use the same dose for the next pain episode or increase to a higher dose following the titration steps described. The successful dose is the one that provides adequate pain relief.*
PRINCIPAL DISPLAY PANEL - 100 mcg Carton - lazanda 16

This is a medication guide for Lazanda, a fentanyl nasal spray used for pain relief. The product is dispensed in 4 bottles containing 8 sprays each and must be administered via nasal administration. Patients must read the enclosed Medication Guide before use. Lazanda is for patients who are tolerant to around-the-clock opioid therapy and must not be substituted for other fentanyl products. The product is sensitive and could be harmful or fatal if given to someone for whom it was not prescribed. Lazanda is available only through a restricted distribution program called the TIRF REMS Access program, and users can contact 1.866822-1483 or TIRFREMSaccess.com for more information.*
PRINCIPAL DISPLAY PANEL - 400 mcg Carton - lazanda 17

This is a medication guide for Lazanda, a fentanyl nasal spray used around the clock for opioid therapy. The enclosed Medication Guide contains important warnings and instructions, and patients must be tolerant of around-the-clock opioid therapy. Lazanda is only available through a restricted distribution program called the TIRF REMS ACCESS program. The prescription must be given with caution, and the patients must be informed about the use of the product including the Verification Guide. The chemical details and dosage recommendations are enclosed, along with information on storage and precautions.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.