Product Images Effer-k 10 Meq Cherry Vanilla
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Effer-k 10 Meq Cherry Vanilla NDC 51801-014 by Nomax Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 10mEq Tablet Pouch Carton - Unflavored - efferk01
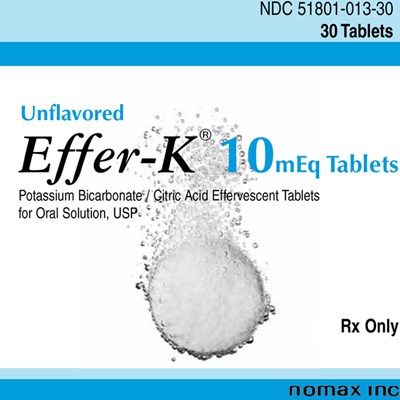
NDC 51801-013-30 is a package of 30 unflavored effervescent tablets known as Effw"-K T OmEq Tablets. These tablets contain Potassium Bicarbonate and Citric Acid and are intended for oral solution. The tablets are USP approved.*
PRINCIPAL DISPLAY PANEL - 10 mEq Tablet Pouch Carton - Cherry Vanilla - efferk02

This is a description of a medication named RINETIE with NDC 51801-014-30. The medication is in the form of effervescent tablets and contains Potassium Bicarbonate and Citric Acid. The flavor of the medication is Cherry Vanilla. The tablets are for oral solution and are available with a prescription only. The manufacturer of the medication is Nomax Inc.*
PRINCIPAL DISPLAY PANEL - 20 mEq Tablet Pouch Carton - Unflavored - efferk03

This is a medication with NDC code 51801-011-30, named RNELIE S, in effervescent tablet form, containing Potassium Bicarbonate and Citric Acid for oral solution. It is intended for prescription use only. The product is unflavored and manufactured by Nomax Inc.*
PRINCIPAL DISPLAY PANEL - 20mEq Tablet Pouch Carton - Orange Cream - efferk04

This is a prescription medicine with the NDC code 51801-012-30. It is an effervescent tablet that can be dissolved in water for oral consumption. The brand name of the medicine is Orange Cream® and it contains potassium bicarbonate and acid. The manufacturer of the medicine is Nomax Inc.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.