Product Images Haloette
View Photos of Packaging, Labels & Appearance
- Figure 1 - haloette 01
- Figure 2 - haloette 02
- Chemical Structure - haloette 03
- Chemical Structure - haloette 04
- Figure 2 - haloette 05
- Chart - haloette 06
- Figure - haloette 07
- Figure A-C - haloette 08
- Figure D-E - haloette 09
- Figure F - haloette 10
- Figure G - haloette 11
- Figure H - haloette 12
- Figure I - haloette 13
- Figure J - haloette 14
- PRINCIPAL DISPLAY PANEL - 3 Ring Pouch Carton - haloette 15
Product Label Images
The following 15 images provide visual information about the product associated with Haloette NDC 51862-148 by Mayne Pharma Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 2 - haloette 02
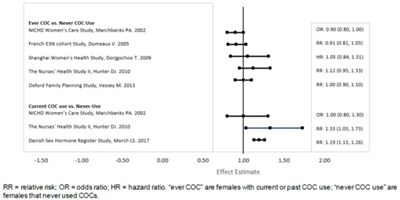
This document provides a list of studies comparing the usage of COC (combined oral contraceptive) in women and its effect on their health. The studies are categorized based on whether the subjects were current or past users of COC (ever COC) or whether they had never used COCs (never COC use). The text also explains the abbreviations used for the three types of effect estimates (RR, OR, and HR).*
Chemical Structure - haloette 04

ETHINYL ESTRADIOL is a synthetic form of the hormone estrogen used in various hormonal contraceptives and in hormone replacement therapy for menopausal women. It works by preventing ovulation and thickening the cervical mucus to block sperm from reaching the egg. It is also sometimes used to treat certain menstrual problems and acne.*
Chart - haloette 06
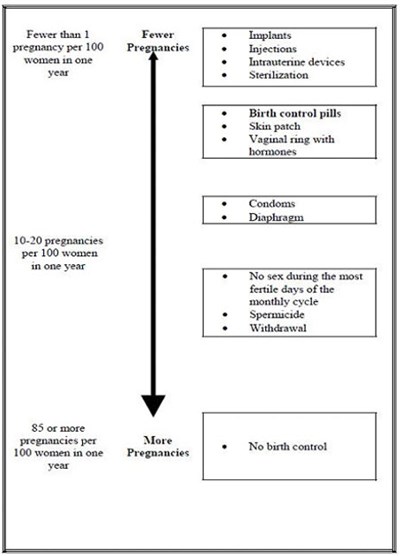
This text provides information regarding various methods of birth control and the corresponding rates of pregnancy. The text suggests that methods such as injections, intrauterine devices, sterilization, birth control pills, skin patch, vaginal ring with hormones, condoms, diaphragm, spermicide, withdrawal, and abstaining from sex during fertile days can all be effective in preventing pregnancy. The text also indicates that the rates of pregnancy can vary depending on the method of birth control, with some methods resulting in fewer than one pregnancy per 100 women, and others resulting in 85 or more pregnancies per 100 women.*
PRINCIPAL DISPLAY PANEL - 3 Ring Pouch Carton - haloette 15

HALOETTE is a vaginal ring contraceptive device that delivers 0.120 mg of etonogestrel and 0.015 mg of ethinyl estradiol per day. It is intended to prevent pregnancy and not protect against HIV or other sexually transmitted diseases. It should be stored refrigerated between 2-8°C before dispensing to the patient, and after dispensing it can be stored at 25°C for up to 6 months. The dispenser should place an expiration date on the adhesive labels provided in the product carton, not exceeding either 6 months from the time of dispensing or the preprinted expiration date if it comes first. The NDC code is 51862-148.03, and it is distributed by Mayne Pharma.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.










