Product Images Galantamine Hydrobromide
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 16 images provide visual information about the product associated with Galantamine Hydrobromide NDC 51990-111 by Yabao Pharmaceutical Co., Ltd. Beijing, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 12 mg Tablet Label - 12mg 60 galantamine

This is a description of a drug called Galantamine Hydrobromide in tablet form, which contains 12mg of Galantamine. It is only available with a prescription, and the dosage information is provided in the accompanying product literature. It is important to keep this drug out of reach of children and dispense it in a tight, light-resistant container as defined in the USP. It needs to be stored at 25°C with permitted excursions to 15°C to 30°C. The lot number and expiration date are provided on the packaging. The drug is distributed by Solco Healthcare US LLC in Cranbury, NJ, and manufactured by Yabao Pharmaceutical Co., Ltd. located in Beijing, China.*
PRINCIPAL DISPLAY PANEL - 4 mg Tablet Label - 4mg 60 galantamine

Galantamine Hydrobromide Tablets are a prescription drug that contains 4mg of galantamine per tablet. The packaging contains 60 tablets with dosage information provided in the accompanying literature. The drug should be kept away from children and stored in a secure container. The medication should be stored at a temperature of 25°C with short-term excursions allowed between 15°C to 30°C. The manufacturer is Solco Healthcare US LLC located in Cranbury, NJ, USA, and it is distributed by Yabao Pharmaceutical Co., Ltd., based in Beijing, China.*
PRINCIPAL DISPLAY PANEL - 8 mg Tablet Label - 8mg 60 galantamine

Galantamine Hydrobromide Tablets, USP containing 8mg of Galantamine hydrobromide in each tablet, supplied in a bottle of 60 tablets. The dosage must be consulted from accompanying product literature. It is an RX only drug and must be kept out of reach of children stored in a tight, light-resistant container under controlled room temperature. It is distributed by Solco Healthcare and manufactured by Yabao Pharmaceutical Co., Ltd in Beijing, China. NDC number for this drug is 51990-112-04.*
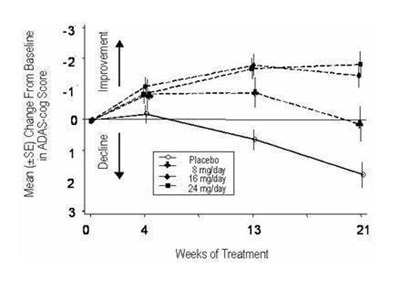
image description - galantamine 06

The text describes a graph or chart showing the cumulative percentage of patients with no change in relation to ADAS-cog (Alzheimer's Disease Assessment Scale - cognitive subscale) change from baseline. The specific data points of the graph or chart, along with additional details about the study or data collection, are not available in the given text.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.