Product Images Oxcarbazepine
View Photos of Packaging, Labels & Appearance
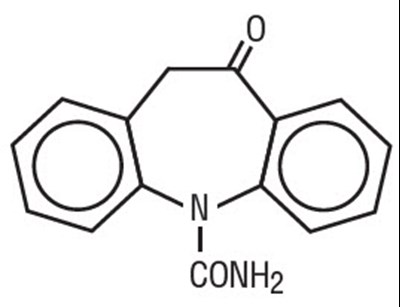
- Chemical Structure - oxcarbazepine 01
- Figure 1 - oxcarbazepine 02
- Figure 2 - oxcarbazepine 03
- Figure 3 - oxcarbazepine 04
- Figure 4 - oxcarbazepine 05
- PRINCIPAL DISPLAY PANEL - 150 mg Tablet Bottle Label - oxcarbazepine 06
- PRINCIPAL DISPLAY PANEL - 300 mg Tablet Bottle Label - oxcarbazepine 07
- PRINCIPAL DISPLAY PANEL - 600 mg Tablet Bottle Label - oxcarbazepine 08
Product Label Images
The following 8 images provide visual information about the product associated with Oxcarbazepine NDC 51991-053 by Breckenridge Pharmaceutical, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - oxcarbazepine 02

This text provides statistical information related to the exit rate based on a log-rank test with a p-value of 0.0001, comparing treatment groups of Oxcarbazepine and Placebo over a trial period from Day 2 to Day 11.*
Figure 2 - oxcarbazepine 03

The given text provides information about the seizure rate study, including the result of the log-rank test with a p-value of 0.046 and a table showing days from the first dose to the first seizure in the treatment groups Oxcarbazepine and Placebo. This data could be related to a clinical trial or a pharmaceutical study.*
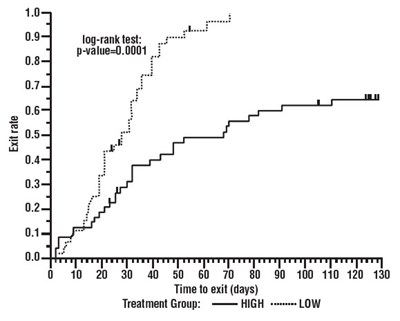
Figure 4 - oxcarbazepine 05

The text provides statistical information on exit rate using the log-rank test with a p-value of 0.0001. The graph displays time to exit in days based on treatment groups, with a notable difference in the exit rate between the treatment groups identified as "Grot" and "HIGH ow".*
PRINCIPAL DISPLAY PANEL - 150 mg Tablet Bottle Label - oxcarbazepine 06

Each film-coated tablet contains 150 mg of oxcarbazepine. It is recommended to refer to the insert for dosage and complete product information. The medication may contain FD&C Yellow No. 5 as a color additive. Store between 20°C-25°C (68°F); excursion permitted to 15°C-30°C (59°F-86°F). Dispense in a tight container. This medication is available in a package of 100 tablets. Distributed by Breckenridge Pharmaceutical, Inc. and manufactured by Towa Pharmaceutical Europe, S.L. It is essential to keep this and all medication out of the reach of children.*
PRINCIPAL DISPLAY PANEL - 300 mg Tablet Bottle Label - oxcarbazepine 07

Each film-coated tablet of oxcarbazepine contains 300 mg of the active ingredient. This medication guide provides important information regarding dosage and product details. The tablets may contain FD&C Yellow No. 5 as a color additive. Storage instructions include keeping the tablets at 20%-25°C (77°F). Oxcarbazepine is dispensed in a tight container as per USP guidelines. The product is distributed by Breckenridge Pharmaceutical, Inc., and manufactured by Towa Pharmaceutical Europe, S.L. It is essential to keep this medication out of the reach of children.*
PRINCIPAL DISPLAY PANEL - 600 mg Tablet Bottle Label - oxcarbazepine 08

Each film-coated tablet contains 600 mg of oxcarbazepine. The dosage information and complete product details can be found in the insert included with the medication. It is advised to print Medication Guides from the website mentioned for more information. The tablets contain FD&C Yellow No. 5 as a color additive and should be stored at 20°-25°C (77°F) with excursions permitted to 15°-30°C (50°-86°F) as per USP guidelines. Dispensing should be done in a tight container. The product is labeled with NDC 51991-055-01 and is manufactured by Towa Pharmaceutical Europe, S.L. in Spain. It is distributed by Breckenridge Pharmaceutical, Inc. in Berkeley Heights, NJ. Remember to keep this medication out of the reach of children.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.