Product Images Estradiol / Norethindrone Acetate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 12 images provide visual information about the product associated with Estradiol / Norethindrone Acetate NDC 51991-474 by Breckenridge Pharmaceutical, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
norethindrone 06

This is a table showing the number of moderate to severe hot flushes over a 12-week period for two groups of participants: one group of 194 people taking Estradiol/Norethindrone Acetate Tablets 0.5 mg/0.1 mg, and one group of 199 people taking a placebo. The table shows data for week 10 and week 12.*
norethindrone 07

This is a medication called Estradiol/Norethindrone Acetate Tablets, with a strength of 1.0mg/0.5mg. The sample size for this medication was 200.*
norethindrone 09
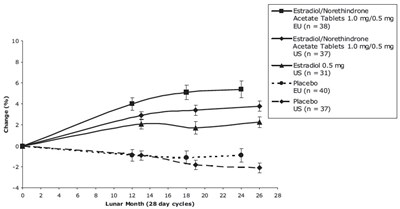
This is a chart showing the change (%) in the effectiveness of different tablets, such as Estradiol/Norethindrone Acetate Tablets 1.0 mg/0.5 mg, Estradiol 0.5 mg, and Placebo, over a period of 5 lunar months (28-day cycles). The data is based on different national samples - EU and US, with varying numbers for each.*
norethindrone 11

This is a description of the packaging for Estradiol / Norethindrone Acetate Tablets, containing 28 tablets with a strength of 1.0 mg / 0.5 mg. The National Drug Code (NDC) for this medication is 51991-474-28. There are various numbers and codes on the packaging, including a prescription designation and a distributor name, but no further information is provided.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.







