Product Images Duloxetine Delayed-release
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 14 images provide visual information about the product associated with Duloxetine Delayed-release NDC 51991-750 by Breckenridge Pharmaceutical, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - duloxetine 02
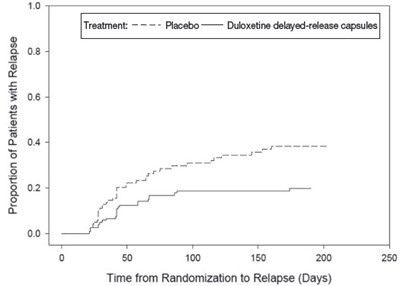
The graph shows the proportion of patients who experienced relapse over time after being randomized for treatment with either placebo or duloxetine delayed-release. The x-axis represents the time in days from randomization to relapse, while the y-axis shows the proportion of patients. The line for duloxetine delayed-release is lower, indicating a lower proportion of patients experienced relapse compared to placebo.*
Figure 2 - duloxetine 03

The text describes a graph that shows the proportion of patients with relapse over time from randomization. The graph compares the placebo group to the group taking duloxetine delayed-release capsules. The x-axis is labeled as "Time from Randomization to Relapse (Days)" with intervals of 50. The y-axis shows the proportion of patients with relapse with intervals of 0.02 from 0 to 0.1.*
Figure 8 - duloxetine 08

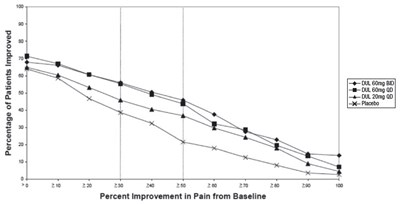
The text describes a graph showing the percentage of patients who improved based on a placebo and Duloxetine (DUL) dosage of 60/120 mg per day. The x-axis displays the percentage of improvement in pain from baseline on a scale of 0 to 100. The y-axis displays the percentage of patients improved on a scale of 50 to 80 for both the placebo and DUL medications.*
Figure 9 - duloxetine 09

This is a chart displaying the percentage of patients who have improved after taking either placebo or DUL 60 mg once daily. The numbers on the left represent the percentage of improvement while the numbers on the bottom represent the dosage or treatment. The chart also displays the percentage improvement in pain from the baseline using the "BOCF" method.*
Figure 10 - duloxetine 10

The chart shows the percentage of patients improved in a clinical trial. The vertical axis represents the percentage of improvement in pain from baseline, and the horizontal axis shows the different treatments (placebo, DULB0/120 mg once daily). There is a significant improvement in pain for patients who received DULB0/120 mg once daily compared to the placebo group. The text "faga" and the numbers at the bottom are not meaningful.*
PRINCIPAL DISPLAY PANEL - 20 mg Capsule Bottle Label - duloxetine 11

Each capsule of this medication contains 22.4 mg of duloxetine hydrochloride USP which is equal to 20 mg duloxetine. The recommended storage temperature is 25°C (77° Fahrenheit) with excursions allowed to 15-30°C (59-86°F). It's suggested to refer to accompanying literature regarding dosage. The delayed-release capsules are available in a container holding 90 capsules. The medication guide should be provided separately to each patient dispensed by the pharmacist. The medicine should be kept in a tightly closed container and out of reach of children. This medication is manufactured in Spain by Towa Pharmaceutical Europe, SL and distributed in the US by Breckenridge Pharmaceutical, Inc. The text contains a Datamatrix code and other information about manufacturing batches and expiry date.*
PRINCIPAL DISPLAY PANEL - 30 mg Capsule Bottle Label - duloxetine 12

Each capsule contains 33.7 mg of duloxetine hydrochloride USP equivalent to 30 mg duloxetine. The medication needs to be stored at room temperature, and the accompanying literature should be consulted for dosage. The medication should be kept in a tightly closed container, out of the reach of children. The label includes information on NDC number and manufacturing company. The product should be dispensed with a medication guide, and the provided space may be used for printing batch and expiration information.*
PRINCIPAL DISPLAY PANEL - 40 mg Capsule Bottle Label - duloxetine 14

This is a medication product that comes in capsules containing 44.9 mg of Duloxetine hydrochloride USP, equivalent to 10-40 mg of Duloxetine. The medication should be stored at temperatures between 15-30 °C (59-86°F). Patients are provided with a guide for dosage which is separate from the medication. It is important to dispense the medication in a tightly closed container and keep it out of reach of children. The manufacturer is Towa Pharmaceutical Europe, S.L. located in Martorell (Barcelona), Spain, while the distributor is Breckenridge Pharmaceutical, Inc. located in Berlin, Connecticut. The product was produced in July 2013 and has an expiration date of September 2020.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.