Product Images Naropin
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 14 images provide visual information about the product associated with Naropin NDC 52584-286 by General Injectables & Vaccines, Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Formula.jpg - Formula1
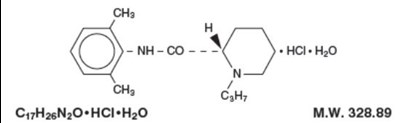
This appears to be a chemical formula and information about its molecular weight. Specifically, the formula is Cy7H26N,0+HCI*H0 with a molecular weight of 328.89. However, without additional context or information, it is difficult to determine the significance or use of this formula.*
Image10.jpg - Image10
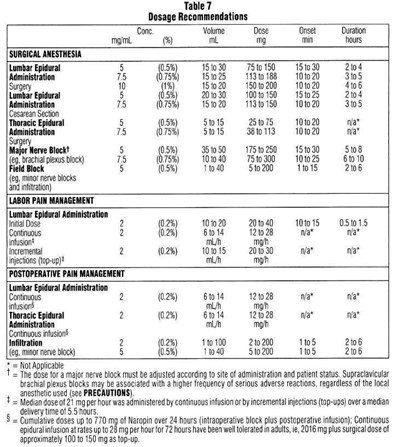
Dosage recommendations for different applications of Naropin (an anesthetic) are given in the text. These include surgical anesthesia, labor pain management, and postoperative pain management. The text provides information on the recommended concentrations, volumes, and doses of Naropin to be administered for epidural administration, nerve block, and infiltration. The onset and duration of the anesthetic effect are also specified for different doses. The text also includes dosage adjustments required based on the patient's status and site of administration. The maximum doses of Naropin for various applications are also given.*
Image11.jpg - Image11
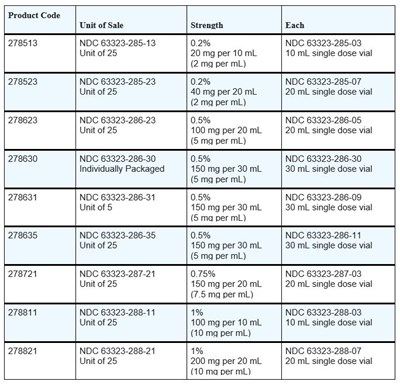
This is a list of different products with unique product codes and unit of sale. The products contain varying strengths of a certain substance per mL and are available in single-dose vials, either individually packaged or in a unit of 5 or 25. The NDC codes for the different products are also included.*
Image12.jpg - Image12
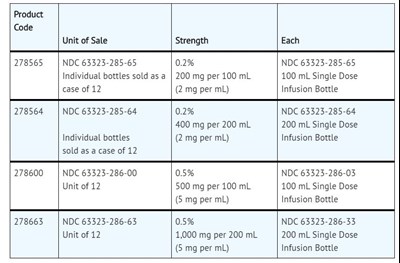
The text describes products with different codes, unit of sale, strength, and Single Dose Infusion Bottle volume. The codes, unit of sale, strength, and Single Dose Infusion Bottle volume are provided for each product.*
Image2.jpg - Image2

This text is a table containing data from a meta-analysis on the mode of delivery during labor and delivery for two medications, Naropin and Bupivacaine. The table shows the number and percentage of deliveries that occurred via spontaneous vertex, vacuum extractor, forceps, and Cesarean section for both medications. Additionally, there is a statistical comparison indicating a significant difference between the two medications.*
Image3.jpg - Image3
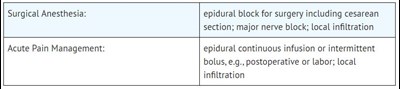
This is a list of surgical anesthesia and acute pain management techniques. For surgical anesthesia, it includes the use of epidural block for cesarean section and major nerve block, as well as local infiltration. For acute pain management, it includes epidural continuous infusion or intermittent bolus, particularly for postoperative or labor cases, as well as local infiltration.*
Image4.jpg - Image4
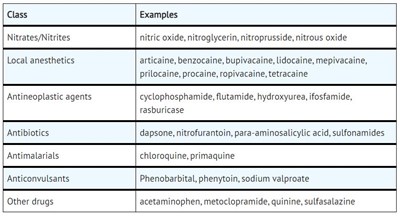
This text provides a list of different classes of drugs, along with specific examples of each class. The drug classes include nitrates/nitrites, local anesthetics, antineoplastic agents, antibiotics, antimalarials, anticonvulsants, and other drugs. Examples of drugs within each class are also listed.*
Image5.jpg - Image5
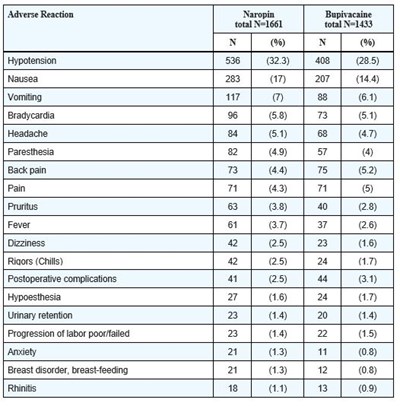
This is a report on the adverse reactions experienced by 1433 patients who were administered bupivacaine. The adverse reactions and their frequencies are listed as follows: Hypotension (85), Nausea (209), Vomiting (117), Bradycardia (58), Headache (68), Paresthesia (82), Back pain (75), Pain (7), Pruritus (63), Fever (61), Dizziness (23), Rigors (Chills) (2), Postoperative complications (61), Hypoesthesia (27), Urinary retention (20), Progression of labor poor/failed (2), Anxiety (2), Breast disorder, breastfeeding (21), and Rnits (18).*
Image6.jpg - Image6
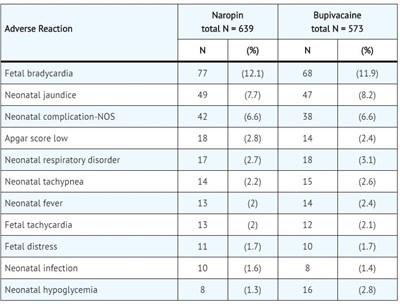
This is a side effect report for the drugs Naropin and Bupivacaine. Adverse reactions were recorded and counted, with the number of occurrences (N) and percentage for each reaction listed. Some of the adverse reactions recorded include fetal bradycardia, neonatal jaundice, and APGAR score low.*
Image7.jpg - Image7
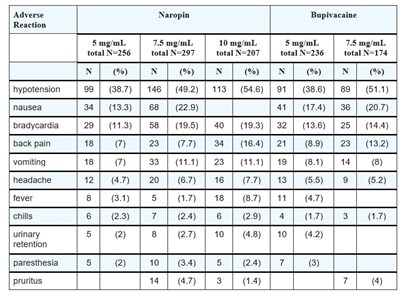
This is a report on adverse reactions to different concentrations of Naropin Bupivacaine. The data shows the number of patients who experienced different symptoms in response to the different concentrations. Symptoms include hypotension, nausea, bradycardia, back pain, vomiting, headache, fever, chills, urinary retention, paresthesia, and pruritus.*
Image8.jpg - Image8
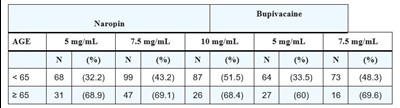
This appears to be a chart showing the percentage of individuals and the number of participants who received certain dosages of Bupivacaine and Naropin based on age. The ages are divided into two categories, those under 65 and those 65 and older. The exact purpose of the chart is unclear without more context.*
Image9.jpg - Image9
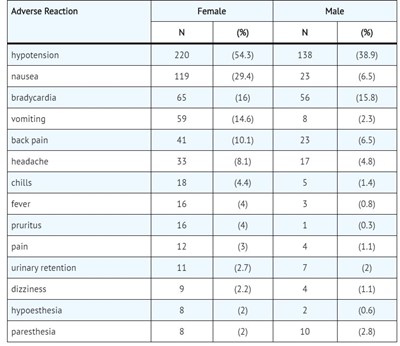
This is a table showing the adverse reactions of a medication in a clinical trial. The reactions are classified by gender, with the number of females and males reporting each reaction as well as the percentage for each group. The adverse reactions range from nausea, vomiting and back pain to fever, pruritus, and urinary retention.*
Label1.jpg - Label1

This is a medical product named Naropin 0.5% (ropivacaine HCL) 150mg/30ml, packaged by General Injectables and Vaccines Inc. It is a single-dose injection with no preservatives intended for infiltration, nerve block, and epidural administration but not for intravenous use. The product should be discarded if the solution is discolored, contains a precipitate or the container closure is not made with natural rubber latex. The manufacturer's insert contains complete product and prescribing information. It is manufactured by Fresenius Kabi with Lot # XXXXXXXXX and NDC # 63323-286-35. The product should be stored between 20 to 26°C (68 to 77°F) in a controlled room temperature and kept out of children's reach.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
