Product Images Clopidogrel
View Photos of Packaging, Labels & Appearance
- clopidogrel-300mg-30s - clopidogrel 300mg 30s
- clopidogrel-300mg-500s - clopidogrel 300mg 500s
- clopidogrel-75mg-1000s - clopidogrel 75mg 1000s
- clopidogrel-75mg-30s - clopidogrel 75mg 30s
- fig1 - clopidogrel figure1
- fig2 - clopidogrel figure2
- fig3-ctd - clopidogrel figure3 ctd
- fig3 - clopidogrel figure3
- fig4 - clopidogrel figure4
- fig5 - clopidogrel figure5
- fig6 - clopidogrel figure6
- fig7 - clopidogrel figure7
- fig8 - clopidogrel figure8
- clopidogrel-structure - clopidogrel structure
Product Label Images
The following 14 images provide visual information about the product associated with Clopidogrel NDC 52605-083 by Polygen Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
clopidogrel-300mg-30s - clopidogrel 300mg 30s

Each film-coated tablet contains 391.5 mg of clopidogrel bisulphate, USP equivalent to 300 mg of clopidogrel. This medication requires a prescription and should be dispensed in a tight, light-resistant container. The container is child-resistant and should be stored at 20°C to 25°C (68°F to 77°F) with permitted excursions between 15°C to 30°C (59°F to 86°F) according to USP Controlled Room Temperature standards. Manufactured by Polygen Pharmaceuticals Inc, this package contains 30 tablets with the National Drug Code 52605-083-13.*
clopidogrel-300mg-500s - clopidogrel 300mg 500s

Each film-coated tablet contains 391.5 mg of clopidogrel bisulphate, equivalent to 300 mg of clopidogrel. It is dispensed in a tight light-resistant container and must be stored at 20°C to 25°C (68°F to 77°F) with excursions permitted between 15°C to 30°C (59°F to 86°F), as stated in the USP Controlled Room Temperature. It's to be dispensed with the accompanying prescribing information and medication guide, and the container should be kept tightly closed. Polygen Pharmaceuticals Inc. in Edgewood, NY 11717 manufactured it. The product has 500 tablets, and the NDC is 52605-083-05.*
clopidogrel-75mg-1000s - clopidogrel 75mg 1000s

This is a medication description for tablets containing 97.875mg of clopidogrel bisulphate, equivalent to 75mg of clopidogrel. The usual dosage information is not available, but it is advised to dispense the medication in a tight, light-resistant container and keep it tightly closed. The tablets should be stored at a temperature between 20°C to 25°C, with excursion permitted between 15°C to 30°C. The manufacturer of the tablets is Polygen Pharmaceuticals Inc. A Medication Guide should be dispensed to each patient.*
fig1 - clopidogrel figure1
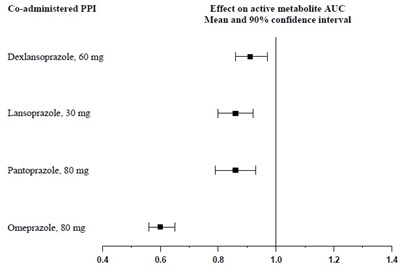
This is a table showing the effect of co-administering different drugs on the active metabolite AUC (area under the curve), with their mean and 90% confidence interval. The drugs listed are Dexlansoprazole (60mg), Lansoprazole (30mg), Pantoprazole (80mg), and Omeprazole (80mg). There is some additional non-textual characters present in the text that do not represent any language, hence have been ignored.*
fig2 - clopidogrel figure2
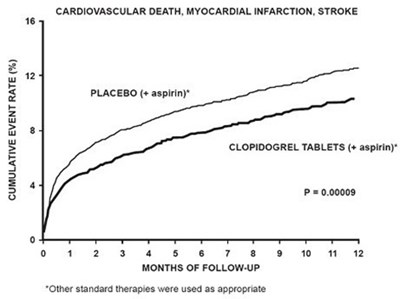
This is a chart showing the cumulative event rate of cardiovascular death, myocardial infarction, and stroke for patients taking a placebo with aspirin versus those taking clopidogrel tablets with aspirin. The chart indicates that the clopidogrel tablets had a notably lower event rate. The chart includes 12 months of follow-up and notes that other standard therapies were used as appropriate.*
fig3 - clopidogrel figure3
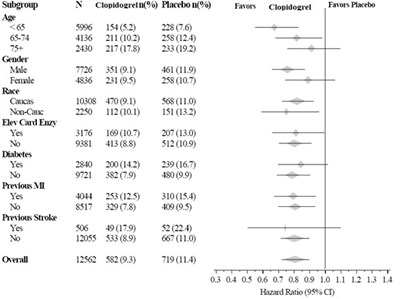
This appears to be a table with statistical data regarding the effects of Clopidogrel versus Placebo on a variety of demographic and health factors such as age, gender, race, and medical history. The table includes subgroup numbers and expected values on a number of variables, as well as a hazard ratio. It is not entirely clear what the overall study was examining or what the findings were, but it appears to be a medical or pharmaceutical study.*
fig4 - clopidogrel figure4
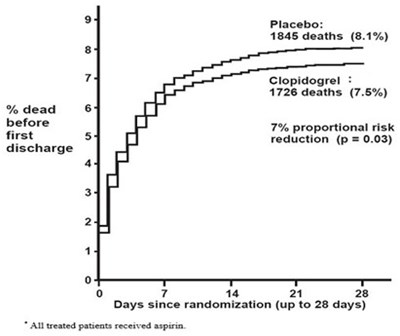
The text describes a comparison between the placebo and Clopidogrel treatments over a period of 28 days. The treatments were compared based on the proportional risk reduction of deaths, with Clopidogrel showing a 7% reduction compared to the placebo. The data is presented on a graph with days since randomization on the X-axis and the proportional risk reduction on the Y-axis.*
fig5 - clopidogrel figure5
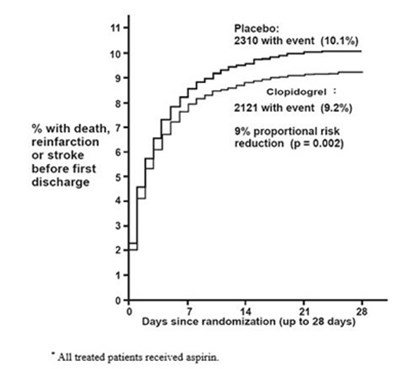
This text describes the results of a study comparing the efficacy of a drug called Clopidogrel with a placebo in reducing the risk of death, reinfarction, or stroke before discharge. The study involved 2310 people who received a placebo and 2121 people who received Clopidogrel. The percentage of people who experienced an event was lower in the Clopidogrel group (9.2%) compared to the placebo group (10.1%). The study found a 9% proportional risk reduction and a p-value of 0.002. The study lasted up to 28 days and all patients received aspci.*
fig6 - clopidogrel figure6
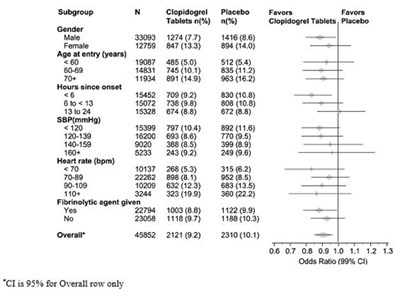
This is a statistical table with information on subgroups, including gender, age at entry, hours since onset, blood pressure, heart rate, and whether the participant received fibrinolytic agent, clopidogrel or a placebo. The table has odds ratio values included but no information on what they represent.*
fig7 - clopidogrel figure7
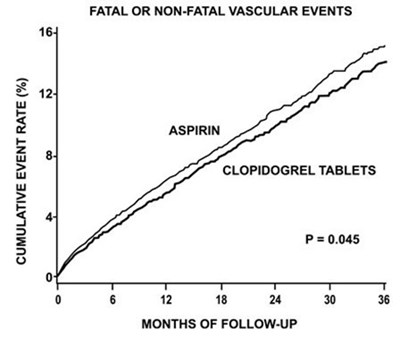
The text represents a chart that shows the cumulative event rate (in percentage) of fatal or non-fatal vascular events with Aspirin and Clopidogrel tablets over a follow-up period of 6,12 and 18 months, respectively. The chart indicates that Clopidogrel tablets have a lower cumulative event rate compared to Aspirin. Additionally, the chart suggests that the difference between the two is considered statistically significant with a P-value of 0.045.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.



