Product Images Emflaza
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 23 images provide visual information about the product associated with Emflaza NDC 52856-503 by Ptc Therapeutics, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
The structural formula for EMFLAZA is deflazacort (a corticosteroid). Corticosteroids are adrenocortical steroids, both naturally occurring and synthetic. The molecular formula for deflazacort is C25H31NO6. The chemical name for deflazacort is (11β,16β)-21-(acetyloxy)-11-hydroxy-2'-methyl-5'H-pregna-1,4-dieno[17,16-d]oxazole-3,20-dione. - emflaza 01
![emflaza-01.jpg The structural formula for EMFLAZA is deflazacort (a corticosteroid). Corticosteroids are adrenocortical steroids, both naturally occurring and synthetic. The molecular formula for deflazacort is C25H31NO6. The chemical name for deflazacort is (11β,16β)-21-(acetyloxy)-11-hydroxy-2'-methyl-5'H-pregna-1,4-dieno[17,16-d]oxazole-3,20-dione. - emflaza 01](https://ndclist.com/assets/spl/images/31b347d2-f156-4055-9d8f-7cf0df420296/400x-emflaza-01.jpg)
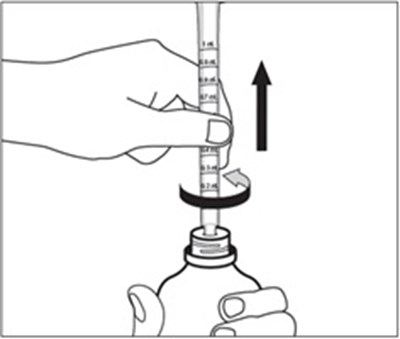
Figure A - emflaza 02
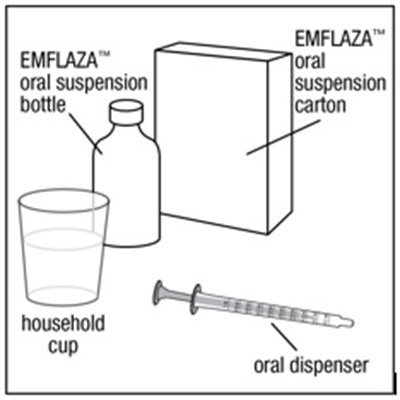
EMFLAZA™ is an oral suspension that comes with an oral dispenser. It may be used in households.*
6 mg Tablet 100-Count Bottle Label - emflaza 14

This is a description of Emflaza® tablets, which come in a bottle with NDC number 52856-501-01 and contains 100 tablets. Each tablet contains 6 mg deflazacort, and the recommended dosage and administration should be consulted in the package insert. The tablets should be stored at 20°C to 25°C (68°F to 77°F). Emflaza® is a registered trademark of PTC Therapeutics, Inc. The text does not include the expiry date or lot number, so it is not-available.*
6 mg Tablet 100-Count Carton Label - emflaza 15
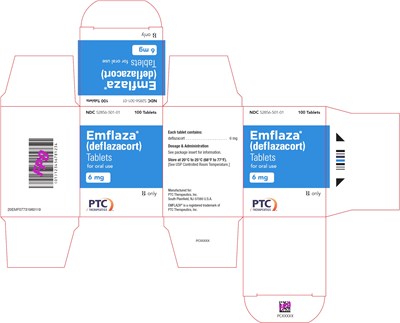
This is a description of a medication called EMFLAZA® manufactured by PTC Therapeutics, Inc. The medication comes in 100 tablet form for oral use and its NDC is 52856-501-01. The tablets contain deflazacorteassnprnRaintanate. Dosage and administration information should be found in the package insert included with the tablets. The medication should be stored at a temperature between 20°C to 25°C (68°F to 77°F). EMFLAZA® is a registered trademark of PTC Therapeutics, Inc.*
18 mg Tablet 30-Count Bottle Label - emflaza 16

This is a medication called Emflaza containing the active ingredient deflazacort, with NDC number 52856-502-03. Each bottle of this medication contains 30 tablets. There is no dosage and administration information provided in this text, and the reader is advised to consult the package insert for this information. This medication should be stored at a temperature between 20°C to 25°C. Emflaza is a registered trademark of PTC Therapeutics, Inc.*
18 mg Tablet 30-Count Carton Label - emflaza 17
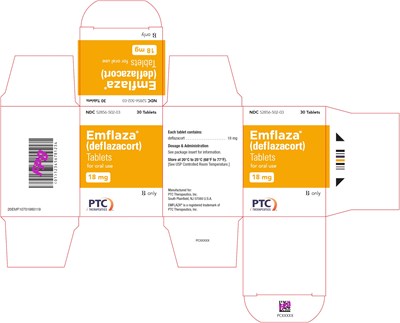
This appears to be information concerning a medication, likely called Emflaza, a registered trademark of PTq. Each tablet contains deflazacort. The dosage and administration details are not available but can be found in the package insert. The medication comes in 30 tablets, and should be stored at 20°C to 25°C (68°F to 77°F). It is for oral use and is manufactured for PTC Therapeutics, Inc. in South Plainfield, NJ 07080 U.S.A.*
30 mg Tablet 30-Count Bottle Label - emflaza 18

This is a medication label for NDC 52856-503-03 containing 30 tablets of Deflazacort, each tablet containing 30 mg of Deflazacort. Dosage and administration information is available in the package insert. Store the medication at a temperature between 20°C to 25°C (68°F to 77°F). EMFLAZA® is a registered trademark of PTC Therapeutics, Inc. The lot number and expiration date are present but not readable in the text.*
30 mg Tablet 30-Count Carton Label - emflaza 19

The text is providing information about a medication called Emflaza. The medication is available in tablet form for oral use and each tablet contains deflazacort. The dosage and administration information can be found in the package insert. The medication is manufactured by PTC Therapeutics and should be stored at controlled room temperature between 20°C to 25°C. The text also mentions the trademark for Emflaza.*
36 mg Tablet 30-Count Bottle Label - emflaza 20

This is a description of a medication whose NDC code is 52856-504-03. It is a bottle of 30 tablets of deflazacort, an oral tablet medication which contains 36mg of deflazacort in each tablet. The dosage and administration information can be found in the package insert provided along. The tablets should be stored at a temperature between 20°C to 25°C (68°F to 77°F). The trademark of this medication is EMFLAZA® and it is registered to PTC Therapeutics, Inc. The lot number is given but the expiration date is not.*
36 mg Tablet 30-Count Carton Label - emflaza 21
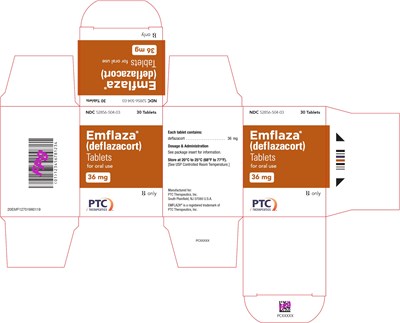
EMFLAZA® tablets are for oral use and each tablet contains 36mg of deflazacort. They are provided in a package with 30 tablets, and the recommended dosage and administration instructions are available in the package insert. The tablets should be stored between 20°C to 25°C (68°F to 77°F) and the USP Controlled Room Temperature guidelines should be followed. EMFLAZA® is a registered trademark of PTC Therapeutics, Inc. The product is manufactured by PTC Therapeutics, Inc. located in South Plainfield, NJ 07080, U.S.A. The product is identified using the NDC 52856-504-03 code.*
22.75 mg/mL Oral Suspension 13 mL Carton Label - emflaza 23

This is a medication called Emflaza, which contains deflazacort. It comes in a press-in bottle adapter for oral suspension administration only. The dosage and administration instructions are included in the package insert. Each mL contains 22.75 mg/m I of deflazacort. The medication should be stored at 20°C to 25°C (68°F to 77°F), and the bottle should be tightly closed after opening. It is manufactured by PTC Therapeutics, Inc. in Spain and is a registered trademark of PTC Therapeutics, Inc.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.