Product Images Nabumetone
View Photos of Packaging, Labels & Appearance
- Structural formula of Nabumetone - 5276f530 14bc 492d 974b 913fd06bf52e 01
- 6MNA - 5276f530 14bc 492d 974b 913fd06bf52e 02
- Table 1 - 5276f530 14bc 492d 974b 913fd06bf52e 03
- 6MNA Plasma Concentrations - 5276f530 14bc 492d 974b 913fd06bf52e 04
- Metabolites - 5276f530 14bc 492d 974b 913fd06bf52e 05
- Table 2 - 5276f530 14bc 492d 974b 913fd06bf52e 06
- Side Effects Table - 5276f530 14bc 492d 974b 913fd06bf52e 07
- Get emergency help if you experience these symptoms - 5276f530 14bc 492d 974b 913fd06bf52e 08
- Stop NSAID if you experience these symptoms - 5276f530 14bc 492d 974b 913fd06bf52e 09
- NSAID medications that need a prescription - 5276f530 14bc 492d 974b 913fd06bf52e 10
- label500 - label
- label750 - labell
Product Label Images
The following 12 images provide visual information about the product associated with Nabumetone NDC 52959-656 by H.j. Harkins Company, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
6MNA - 5276f530 14bc 492d 974b 913fd06bf52e 02

6-methoxy-2-naphthylacetic acid (6MNA) is a chemical compound with the molecular formula COOH-CH40.*
6MNA Plasma Concentrations - 5276f530 14bc 492d 974b 913fd06bf52e 04

This is a graph displaying Nabumetone Active Metabolite (6MNA) Plasma Concentrations at Steady State Following Once-Daily Dosing of Nabumetone. The doses tested were 1000 mg and 2000 mg, with 31 and 12 participants respectively. The x-axis represents TIME (hours) and the y-axis represents plasma concentration values. There are lines showing the mean and standard deviation values for each dose.*
Metabolites - 5276f530 14bc 492d 974b 913fd06bf52e 05

This appears to be a list of different forms and percentages of metabolites (substances formed during metabolism) of nabumetone and its derivatives, including some that are unchanged and others that are conjugated (combined with another substance). The different forms include 6-methoxy-2-naphthylacetic acid (6MNA), 6-hydroxy-2-naphthylacetic acid (6HNA), and 4-(6-hydroxy-2-naphthyl)-butan-2-ol. The percentages indicate the amount of each form as a percentage of the total dose, with some forms being very low or not detectable.*
Table 2 - 5276f530 14bc 492d 974b 913fd06bf52e 06
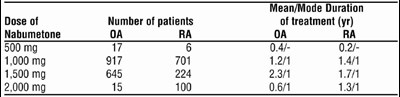
This appears to be a table showing the mean and mode durations of treatment with Nabumetone at different doses for patients with RA (rheumatoid arthritis). The "Dose of treatment" column shows the different amounts of Nabumetone administered (500 mg, 1000 mg, etc.), and the "Number of patients" column indicates how many patients received each dose. The subsequent columns show the mean and mode duration of treatment for each dose in years.*
Side Effects Table - 5276f530 14bc 492d 974b 913fd06bf52e 07

This text provides a description of the serious and other side effects associated with the consumption of a certain medication/drug. The serious side effects include heart attack, stroke, high blood pressure, heart failure, kidney problems, bleeding and ulcers, low red blood cells, life-threatening skin and allergic reactions, liver problems, and asthma attacks. Other side effects include stomach pain, constipation, diarrhea, gas, heartburn, nausea, vomiting, and dizziness.*
Get emergency help if you experience these symptoms - 5276f530 14bc 492d 974b 913fd06bf52e 08

The given text appears to be a list of symptoms with bullet points. It suggests the symptoms of shortness of breath, slurred speech, chest pain, swelling of the face or throat, and weakness in one part or side of the body. These symptoms can be associated with various medical conditions and require medical attention.*
Stop NSAID if you experience these symptoms - 5276f530 14bc 492d 974b 913fd06bf52e 09

This appears to be a list of symptoms that an individual may experience as signs of a medical condition. These symptoms include nausea, fatigue or weakness, itching, yellowing of the skin or eyes, stomach pain, flu-like symptoms, vomiting blood, blood in the stool or black, tarry stool, unusual weight gain, skin rash or blisters with fever, and swelling of the extremities. These symptoms may indicate a serious medical issue and medical attention should be sought if they persist or worsen.*
NSAID medications that need a prescription - 5276f530 14bc 492d 974b 913fd06bf52e 10

This is a list of generic names and corresponding brand names of various pain medications. The medications listed include nonsteroidal anti-inflammatory drugs (NSAIDs) such as Celecoxib (Celebrex), Diclofenac (Cataflam, Voltaren, Arthrotec), among others. It also includes COX-2 inhibitors, including Celecoxib (Celebrex), and various other medications, including Mefenamic Acid (Ponstel), Meloxicam (Mobic), Nabumetone (Relafen), and Piroxicam (Feldene), among others.*
label500 - label

This is a description of a medication called Nabumetone 500mg Tablet. It is manufactured by Glenmark and comes in a lot number NBT500KG with an expiration date of 10/10. The pill is oval-shaped, white, and film-coated. It should be taken as directed by a doctor or according to the dosage information on the outsert. There is a caution statement that advises against abusing the medication, and it should be kept in a container at room temperature. The text also mentions that Nabumetone is comparable to Relafen, which has the same pill ID number as Nabumetone and comes in a lot number 68462-358-05.*
label750 - labell
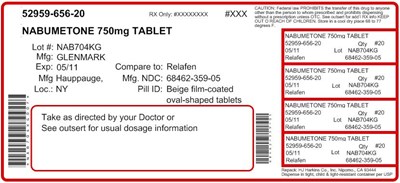
This is a medication description for a NABUMETONE 750mg TABLET. The tablets are oval and beige film-coated. It is advised to take them as directed by a doctor or check the enclosed insert for dosage details. The medication is manufactured by Glenmark, and the NDC for the manufacturing location is 68462-359-05. The lot number for the tablets is NAB704KG, and the expiry date is 05/11. Contains lot details for Gagrast and a warning message indicating that it is prohibited to transfer the tablets.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

