Product Images Lyrica
View Photos of Packaging, Labels & Appearance
- PRINCIPAL DISPLAY PANEL - 50 mg Capsule Bottle - label1
- PRINCIPAL DISPLAY PANEL - 75 mg Capsule Bottle - label2
- PRINCIPAL DISPLAY PANEL - 100 mg Capsule Bottle - label3
- PRINCIPAL DISPLAY PANEL - 150 mg Capsule Bottle - label4
- Figure - lyrica 01
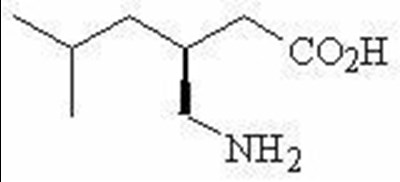
- Chemical Structure - lyrica 02
- Figure - lyrica 03
- Figure - lyrica 04
- Figure - lyrica 05
- Figure - lyrica 06
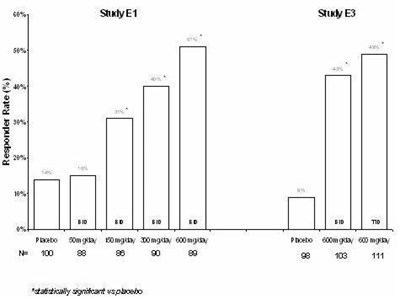
- Figure - lyrica 07
- Figure - lyrica 08
- Figure - lyrica 09
- Figure - lyrica 10
- Figure - lyrica 11
- Logo - lyrica 12
Product Label Images
The following 16 images provide visual information about the product associated with Lyrica NDC 52959-891 by H.j. Harkins Company, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PRINCIPAL DISPLAY PANEL - 50 mg Capsule Bottle - label1

This is a label of a medication named LYRICA 50mg capsule manufactured by Pfizer. It is a prescription drug and should be used as directed by a doctor. The lot number of the capsule is LYR510PZ, and it was manufactured in Vega Baja, Puerto Rico. The expiration date of the product is 11/12. The NDC number for this capsule is 52959-891-30, and the pill ID is a white round capsule with the number 0071-1013-68. The box contains 30 capsules. The rest of the text seems to be a repetition of this information with some variations and is not very useful.*
PRINCIPAL DISPLAY PANEL - 75 mg Capsule Bottle - label2

This is the label of a bottle of Lyrica 75mg capsules manufactured by Pfizer in Vega Baja, PR with lot number LYCO5PZ and expiration date 08/10. The Rx number is 000000. The pills are white/orange capsules and are only available with a prescription. The label provides caution regarding the transfer of the pill and instructs patients to take as directed by their doctor or to see the enclosed leaflet for usual dosage information. Other text on the label appears to be illegible or a repetition of the information provided.*
PRINCIPAL DISPLAY PANEL - 100 mg Capsule Bottle - label3

This is the packaging information of a medication called LYRICA 100mg Capsule, manufactured by Pfizer and located in Vega Baja, PR. The lot number is LIR104PZ and the expiration date is 10/10. The prescription is only allowed with a valid RX and the usual dosage information can be found on the outsert. The remaining text is not clear and includes gibberish characters.*
PRINCIPAL DISPLAY PANEL - 150 mg Capsule Bottle - label4

This is a description of a prescription drug called Lyrica. The package contains 30 white round capsules with strength of 150mg each. The manufacturer is Pfizer and the lot number is LRC155PZ. It should be taken as directed by a doctor with usual dosage information available on the insert. The text includes a medication guide with the National Drug Code (NDC) 0071-1016-68 and expiration date of 04/10. No information is available about the other text "52959-897-30 RX Only: 10000000 KKK e e e A o e U REREHTBF EABREN Sire i co oy pioce 5107" as it seems unrelated.*
Figure - lyrica 01

This appears to be a medical formula for estimating creatinine clearance for female patients. The formula takes into account the patient's age, weight in kilograms, and serum creatinine levels in mg/dL. However, it is difficult to fully understand the formula without additional context or information.*
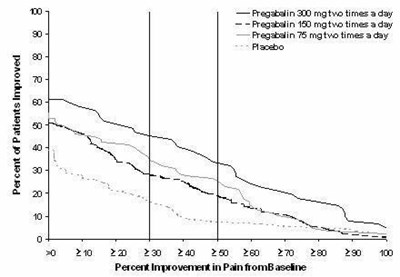
Figure - lyrica 07

This appears to be a chart or table, displaying information about the percentage of patients who improved with different treatments. It includes the medication Pregatalin (at a dosage of 100 mg and a frequency of 3 times per day), as well as Pregabalin (at an unspecified dosage but a frequency of 3 times per day), and a placebo (called "Placeta"). The last line seems to show the percentage improvement in pain from a baseline measurement. However, the rest of the text is not legible or does not appear to be in English.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.