Product Images Metformin Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Metformin Hydrochloride NDC 53002-1251 by Rpk Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
bd3a3e6-1b64-4bda-88db-ae433d17dc8a - 0bd3a3e6 1b64 4bda 88db ae433d17dc8a
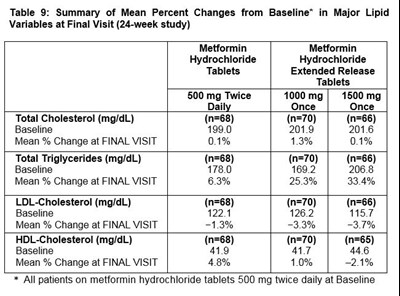
The table shows the summary of mean percent changes from Baseline in major lipid variables at the final visit of a 24-week study. The study involved patients on Metformin Hydrochloride Tablets Extended Release of different doses once or twice a day. The changes indicate the percentages of change from baseline for different lipid variables, such as total cholesterol, total triglycerides, LDL-cholesterol, and HDL-cholesterol. Patients on different doses of the medication experienced varying changes from baseline during the course of the study.*
705cc8a-5055-412c-825a-80a3fd5b9c7a - 3705cc8a 5055 412c 825a 80a3fd5b9c7a

The given text shows a table that displays the mean changes in HbAs, Fasting Plasma Glucose, and Body Weight at Week 12 and the final visit of a 24-week study. The study compares the changes observed in patients who were given either Metformin Hydrochloride 500mg twice daily, Metformin Hydrochloride 1000mg once daily or Metformin Hydrochloride 1500mg once daily. The table displays the baseline, changes at 12 weeks and the final visit, 95% confidence intervals, and the number of patients (n) for each dosage. There is not enough information to determine the purpose or findings of the study.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.



