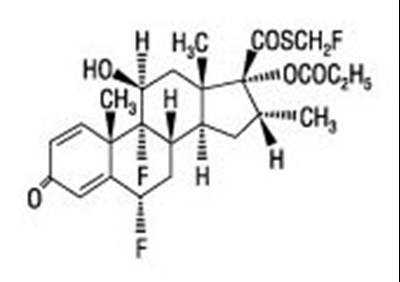
Product Images Fluticasone Propionate And Salmeterol Diskus
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 16 images provide visual information about the product associated with Fluticasone Propionate And Salmeterol Diskus NDC 53002-1568 by Rpk Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1. Mean Percent Change from Baseline in FEV1 in Subjects with Asthma Previously Treated with Either Inhaled Corticosteroids or Salmeterol (Trial 1) - fp sal spl graphic 03
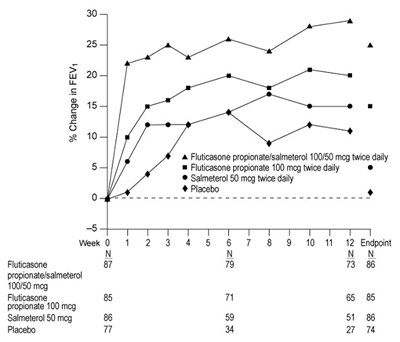
Figure 2. Percent Change in Serial 12-Hour FEV1 in Subjects with Asthma Previously Using Either Inhaled Corticosteroids or Salmeterol (Trial 1) - fp sal spl graphic 04

Figure 3. Percent Change in Serial 12-Hour FEV1 in Subjects with Asthma Previously Using Either Inhaled Corticosteroids or Salmeterol (Trial 1) - fp sal spl graphic 05

Figure 4. Predose FEV1: Mean Percent Change from Baseline in Subjects with Chronic Obstructive Pulmonary Disease - fp sal spl graphic 06