Product Images Lutera
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Lutera NDC 53002-1632 by Rpk Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
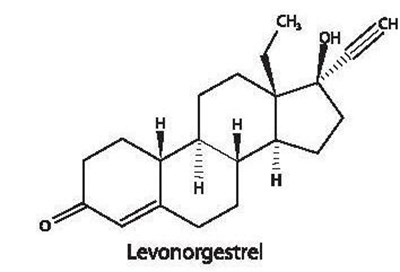
Chemical Structure - lutera 02
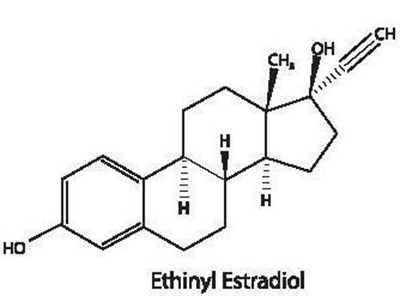
Ethinyl Estradiol is a synthetic estrogen used in combination with a progestin as an oral contraceptive. It is also used in the treatment of menopausal symptoms such as hot flashes, vaginal dryness, and osteoporosis. It works by preventing ovulation and changing the cervical mucus and uterine lining, making it more difficult for sperm to reach the uterus and for a fertilized egg to attach to the uterus.*
Figure I - lutera 03
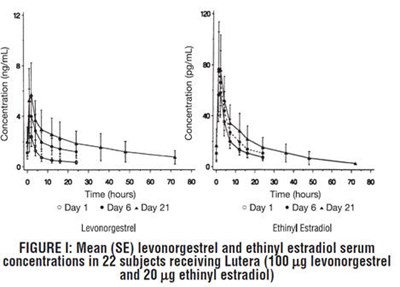
This is a graph showing the mean levonorgestrel and ethinyl estradiol serum concentrations in 22 subjects receiving Lutera 50 g levonorgestrel and 20 g ethinyl estradiol over the course of 21 days. There is no additional information available from the given text.*
Figure II - lutera 04
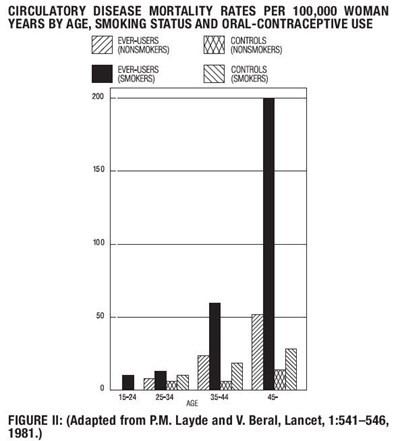
The text describes a chart showing circulatory disease mortality rates per 100,000 woman years categorized by age, smoking status, and oral-contraceptive use. The chart includes data for ever-users controls for non-smokers and smokers. The chart contains a scale from 0 to 150. The source of the chart is Lancet, volume 1, pages 541-546 in 1981.*
image - lutera 05
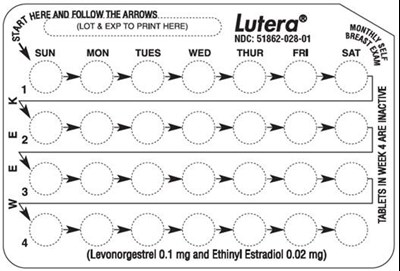
This is a medication label for Lutera® birth control pills. It includes the National Drug Code (NOC) and indicates that the tablets in week 4 are inactive. The label also instructs the user to follow the arrows when taking the pills.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.