Product Images Losartan Potassium
View Photos of Packaging, Labels & Appearance
- Figure 1 - 92296151 e507 41b4 a040 c01b87ecb982 01
- figure 2 - 92296151 e507 41b4 a040 c01b87ecb982 02
- figure 3 - 92296151 e507 41b4 a040 c01b87ecb982 03
- figure 4 - 92296151 e507 41b4 a040 c01b87ecb982 04
- figure 5 - 92296151 e507 41b4 a040 c01b87ecb982 05
- figure 6 - 92296151 e507 41b4 a040 c01b87ecb982 06
- figure 6 - 92296151 e507 41b4 a040 c01b87ecb982 07
- Label Image - lbl530022643
Product Label Images
The following 8 images provide visual information about the product associated with Losartan Potassium NDC 53002-2643 by Rpk Pharmaceuticals, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
figure 2 - 92296151 e507 41b4 a040 c01b87ecb982 02

This appears to be a graphical representation of data related to a study on the effectiveness of Atenolol and Losartan Potassium Tablets in reducing risk for some sort of primary endpoint. The data shows an "Adjusted Risk Reduction" of 13% with a p-value of 0.021. The x-axis appears to represent study months, while the y-axis may represent the percentage of patients with the primary endpoint. Without additional context or information, it is difficult to determine the exact nature of the study or the primary endpoint.*
figure 3 - 92296151 e507 41b4 a040 c01b87ecb982 03
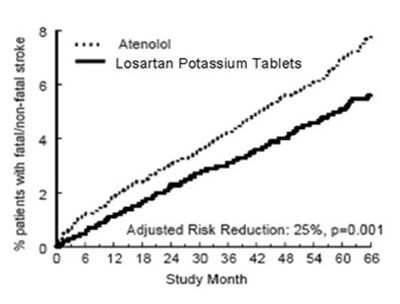
The text appears to be a result comparison between Atenolol and Losartan Potassium Tablets for patients with fatal/non-fatal stroke indicating an adjusted risk reduction of 25% with a p-value of 0.001 over a period of study months ranging from 6 to 68.*
figure 5 - 92296151 e507 41b4 a040 c01b87ecb982 05

This appears to be a table showing the percentage of patients with an event and some calculations for risk reduction. There is also a mention of losartan potassium tablets and a placebo, which may be related to the numbers in the table, but without more information it's difficult to know for sure.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.




