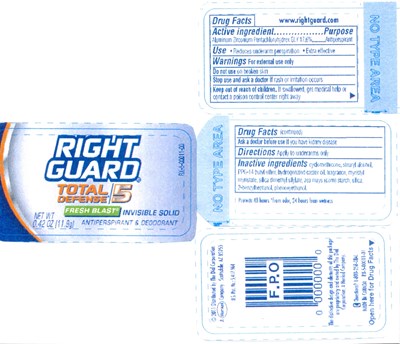
FDA Label for Rg Td5 Inv.solid Antiperspirant Deodorant Fresh Blast
View Indications, Usage & Precautions
Rg Td5 Inv.solid Antiperspirant Deodorant Fresh Blast Product Label
The following document was submitted to the FDA by the labeler of this product The Dial Corporation. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Otc - Active Ingredient
Aluminum Zirconium Pentachlorohydrex Gly 17.8%
Otc - Purpose
Antiperspirant
Protects 48 hours from odor, 24 hours from wetness.
Indications & Usage
Use: Reduces underarm perspiration , Extra effective
Warnings
Warnings: For external use only.
Otc - Do Not Use
Do not use on broken skin.
Otc - Stop Use
Stop use and ask a doctor if rash or irritation occurs.
Otc - Keep Out Of Reach Of Children
Keep out of reach of children. If swallowed, get medical help or contact a poison control center right away.
Otc - Ask Doctor/Pharmacist
Ask a doctor before use if you have kidney disease.
Dosage & Administration
Directions: Apply to underarms only.
Inactive Ingredient
Cyclomethicone, stearyl alcohol, PPG-14 butyl ether, hydrogenated castor oil, fragrance, myristyl myristate, silica dimethyl silylate, zea mays (com) starch, silica, 2-benzylheptanol, phenoxyethanol.
How Supplied
2011 Distributed by The Dial Corporation A Henkel Company Scottsdale, AZ, 85255. The distinctive design and elements of this package are proprietary and owned by the Dial Corporation, a Henkel Company. U.S.PAT.NO.:5,417,964
Otc - Questions
Questions? 1-800-258-DIAL, MADE IN CANADA
Package Label.Principal Display Panel
* Please review the disclaimer below.