FDA Label for Spf 15 Warm Tan
View Indications, Usage & Precautions
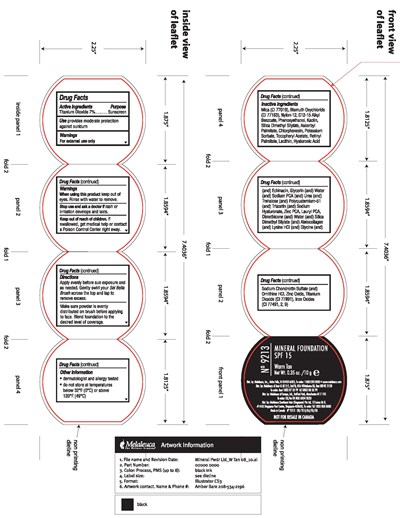
Spf 15 Warm Tan Product Label
The following document was submitted to the FDA by the labeler of this product Melaleuca, Inc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Dosage & Administration
Directions
Apply evenly before sun exposure and as needed. Gently swirl your Sei Bella Brush across the top and tap to remove excess.
Make sure powder is evenly distributed on brush before applying to face. Blend foundation to the desired level of coverage.
Clinical Studies
Other Information
- dermatologist and allergy tested
Storage And Handling
Other Information
- do not store at temperatures below 32°F (0°C) or above 120°F (49°C)
Inactive Ingredient
Enter section text here
Package Label.Principal Display Panel
Otc - Active Ingredient
Active ingredients Purpose
Titanium Dioxide...............Sunscreen
* Please review the disclaimer below.