Product Images Lotronex
View Photos of Packaging, Labels & Appearance
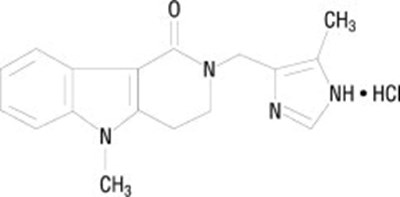
- Chemical Structure - lot00 0006 01
- Figure 1 - lot00 0006 02
- Principal Display Panel - Lotronex 0.5 mg Bottle Label - lot00 0007 03
- Principal Display Panel - Lotronex 0.5 mg Carton - lot00 0007 04
- Principal Display Panel - Lotronex 1 mg Bottle Label - lot00 0007 05
- Principal Display Panel - Lotronex 1 mg Carton - lot00 0007 06
Product Label Images
The following 6 images provide visual information about the product associated with Lotronex NDC 54766-895 by Sebela Pharmaceuticals Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Principal Display Panel - Lotronex 0.5 mg Bottle Label - lot00 0007 03

This is a medication guide for Lotronex (alosetron hydrochloride) tablets, dispensed in a package of 30 tablets. Each film-coated tablet contains alosetron hydrochloride equivalent to 0.5 mg alosetron. The guide includes prescribing information for dosage and administration. The NDC number is 54766-894-03, and it complies with Federal law. The document is intended for healthcare professionals and patients who need information about the medication.*
Principal Display Panel - Lotronex 0.5 mg Carton - lot00 0007 04

This is a description of a medication called Lotronex. It comes in the form of 30 film-coated tablets, each containing a drug called alosetron hydrochloride equivalent to 0.5mg of the active ingredient.*
Principal Display Panel - Lotronex 1 mg Bottle Label - lot00 0007 05

This appears to be a description of a medication called "LOTRONEX", which is a type of tablet. It contains the active ingredient alosetron hydrochloride and is available in 1mg strength. This medication is regulated by federal law and must be accompanied by a medication guide when dispensed. The text also provides contact information for the distributor, Sebela Pharmaceuticals.*
Principal Display Panel - Lotronex 1 mg Carton - lot00 0007 06

This is a description of two different medications called LOTRONEX and LUTHUNEX. The medications come in tablet form and each film-coated tablet contains either alosentron hydrochloride or dosentron hydrochloride. Both medications have a strength of 1mg. The price of 30 tablets is shown as "£".*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.