Product Images Fluticasone Propionate
View Photos of Packaging, Labels & Appearance
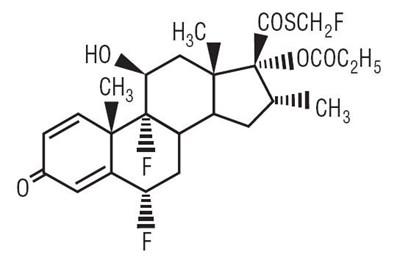
- structural formula image - 260483b0 a145 4655 9d44 1d3069551c4d 01
- Table 1 - 260483b0 a145 4655 9d44 1d3069551c4d 02
- Table 2 - 260483b0 a145 4655 9d44 1d3069551c4d 03
- Table 3 - 260483b0 a145 4655 9d44 1d3069551c4d 04
- Table 4 - 260483b0 a145 4655 9d44 1d3069551c4d 05
- Table 5 - 260483b0 a145 4655 9d44 1d3069551c4d 06
- Table 6 - 260483b0 a145 4655 9d44 1d3069551c4d 07
- image of package label - 5337
Product Label Images
The following 8 images provide visual information about the product associated with Fluticasone Propionate NDC 54868-5337 by Physicians Total Care, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Table 1 - 260483b0 a145 4655 9d44 1d3069551c4d 02

The given text is a table showing various drug-related adverse events related to the skin for patients who were given Fluticasone Once, Fluticasone Twice, and Vehicle Twice. The table has details about the number of patients observed for the drug effect and the type of skin reaction. The adverse events include skin infections, eczema, viral warts, herpes simplex, impetigo, atopic dermatitis, exacerbation of eczema, erythema, burning, stinging, skin irritation, pruritus, exacerbation of pruritus, folliculitis, blisters, and dryness of the skin.*
Table 2 - 260483b0 a145 4655 9d44 1d3069551c4d 03

Table 2 summarizes the adverse events encountered in a pediatric open-label trial where Fluticasone was administered twice a day. The table includes adverse events such as Buting, Dusky Erythema, Erythematous rash, Fadal and Nor-fadal telangiectasia, and Urticaria. For additional details, one may refer to the accompanying text. The number of participants in the trial was 51.*
Table 3 - 260483b0 a145 4655 9d44 1d3069551c4d 04

This text is a table showing the physician's assessment of clinical response to Fluticasone propionate cream. It includes percentages of patients who are rated as having excellent, good, fair, poor, or worse response based on their physician's assessment.*
Table 4 - 260483b0 a145 4655 9d44 1d3069551c4d 05

This appears to be a table showing improvements in clinical signs with the use of fluticasone propionate cream compared to a vehicle (likely a placebo cream). The clinical signs being measured appear to be Erythema, Tackling, and Scaling. However, further context is needed to understand the significance of the values.*
Table 5 - 260483b0 a145 4655 9d44 1d3069551c4d 06

This appears to be a table showing the physician's assessment of clinical response to two different creams that contain the medication fluticasone propionate, with different dosing instructions. The table includes four columns labeled Study 1 and Study 2 for each of the two creams, with sample sizes for each column. The rows indicate the level of response, ranging from Excellent to Poor or Worse. The percentage of patients in each cell corresponds to the proportion of patients who received that rating for that dosing regimen in that study.*
Table 6 - 260483b0 a145 4655 9d44 1d3069551c4d 07

This is a table that provides mean improvements over baseline for clinical signs and symptoms from two studies using different strengths of Futicasone Propionate cream. The table shows improvements in Erythema, Pruritus, Thickening, Lichenification, Vesiculation and Crusting. The first column mentions a 0.05% cream, applied once daily, and the second column mentions a 0.05% cream, applied twice daily. The table contains four studies (Stdy 1, Sty 2, Study T, Study 2).*
image of package label - 5337

This appears to be a package label for Fluticasone 6.65% cream, distributed by Physicians Total Care with batch number 0060 and expiration date 06/18. The NDC (National Drug Code) number is also listed as 54868-5337-0. However, the "I S3370360500" text is not clear and its meaning is unclear.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.