Product Images Enjuvia
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Enjuvia NDC 54868-6165 by Physicians Total Care, Inc., such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
structural formula for sodium 17 alpha dihydroequilin sulfate - b7aba0b2 72d1 4429 bb64 98b92e9d9433 03
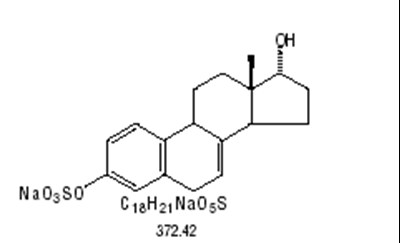
structural formula for for sodium 17 beta dihydroequilin sulfate - b7aba0b2 72d1 4429 bb64 98b92e9d9433 05

structural formula for sodium 17 alpha dihydroequilenin sulfate - b7aba0b2 72d1 4429 bb64 98b92e9d9433 06










