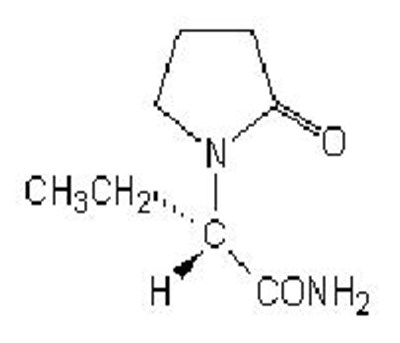
Product Images Levetiracetam
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 13 images provide visual information about the product associated with Levetiracetam NDC 55111-182 by Dr. Reddy's Laboratories Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Carton2 - Carton2
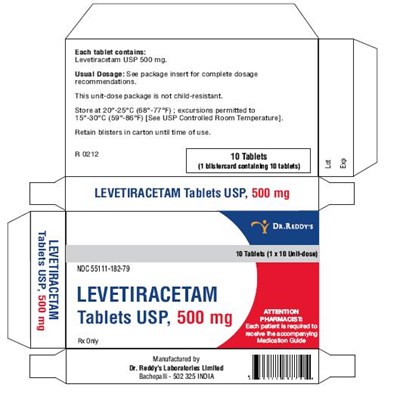
This is a medication label for Levetiracetam Tablets USP, available in 500mg dosage. It contains instructions for storage and dosage, and warns against improper handling. It includes the manufacturer's contact information.*
Carton3 - Carton3
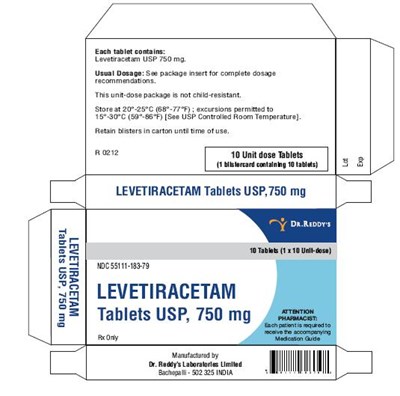
These are Levetiracetam Tablets USP, 750 mg. Each tablet blister pack contains 10 unit dose tablets. The usual dosage is mentioned in the package insert. The uni-dose packaging is crush-resistant and should be stored at controlled room temperature. Any excursions permitted to TEVIS0°C. The product should be kept in its original blister pack and carton.*
Container1 - Container1

This appears to be a medication label for Levetiracetam Tablets USP of 250mg strength. The label indicates that each patient is required to receive a medication guide and there are 120 tablets in the package. The remaining text seems to be gibberish or not in English and may have been errors.*
figure5 - figure5
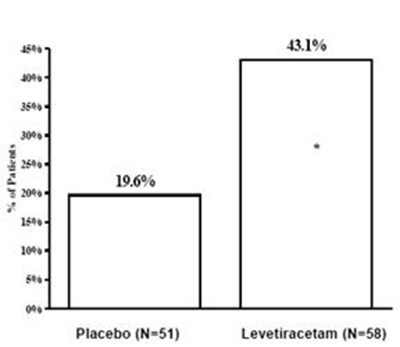
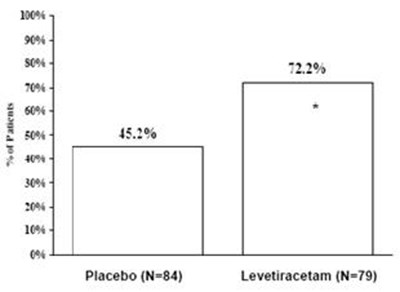
This appears to be a table showing the percentage values for "P" and "196%" for Placebo and Levetiracetam. It is not clear what P and 196% represent. Additionally, the table shows that there were 51 participants in the Placebo group and 58 participants in the Levetiracetam group. Further context is needed to understand the significance of these values.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.