Product Images Venlafaxine Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 11 images provide visual information about the product associated with Venlafaxine Hydrochloride NDC 55111-548 by Dr. Reddy's Laboratories Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
carton4

Each tablet of Venlafaxine Tablet USP contains 75 mg of Venaflaxine Hydrochloride USP. The recommended dosage and storage conditions are given on the package. The package is not child-resistant, and the blister should be retained near the time of use. This medicine comes in a single-dose pack. The NDC number is 5511154879. The product is dispensed with a medication guide provided separately, and it requires a prescription for dispensing.*
carton5
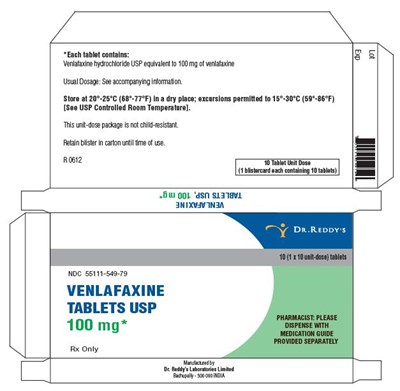
Each tablet contains 100mg of venlafaxine hycochoide USP equivalent of verlatarine. The standard dosage is specified in the accompanying nomination. The tablets should be stored at a temperature of 20°C to 25°C (68°F-77°F) in a dry place. The temperature excursion allowed is 15°C-30°C (59°F-86°F), which is a controlled room temperature. The package is not tamper-evident. The package of this tablet is available in a carton of 5 units. This is a prescription drug with a strength of 100 mg of Venlafaxine in each tablet, and the product is available for sale only under a physician's prescription.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.








