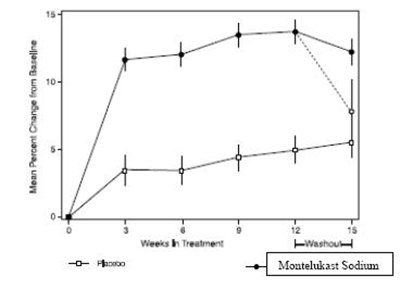
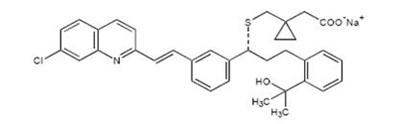
Product Images Montelukast Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 9 images provide visual information about the product associated with Montelukast Sodium NDC 55111-594 by Dr.reddy's Laboratories Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10mgcarton - 10mgcarton

Each tablet contains 10.4 mg Montelukast Sodium USF, equivalent to 10 mg Montelubast. This medication should be stored at room temperature (20°C to 25°C), protected from both moisture and light. There is no recommended dosage, except to see the accompanying circular. These tablets are not child-resistant and should not be dispensed to households. This medication is for institutional use only. The pack contains 100 tablets of Montelukast Sodium, USP, 10mg designed for adults 15 years and older. This product was distributed by Dr. Reddy's Laboratories Inc. from Princeton, NJ, 08540 and was made in India.*
10mgcontainer90 - 10mgcontainer90

This is a description of a medicine called "Sodium Tablets, USP 10 mg" by Dr. Reddy's. It is indicated for adults of age 15 years and older. The tablets are available in a pack of 90 and require a prescription. This medicine helps in the treatment of headaches and other related conditions. The text also contains some irrelevant characters which can be ignored.*
4mgcarton - 4mgcarton

This is a medication label for Montelukast Sodium Chewable Tablets, 4 mg, manufactured by Dr. Reddy's Laboratories. The label also contains an address and zip code for the manufacturer's location. There is also an unidentifiable line of text at the beginning.*
4mgcontainer - 4mgcontainer

This is a description of medication packaging and usage instructions for pediatric patients. The medication is Montelukast Sodium and it comes in chewable tablet form. The package contains 90 tablets and is intended for patients aged 2 to 5 years old. The package includes a patient information leaflet that can be accessed by scanning a QR code or reading it from a paper copy provided by the pharmacist during dispensing. The packaging also includes a National Drug Code (NDC) 55111-693.90 and was created for prescription use only.*
5mgcontainer - 5mgcontainer

This appears to be a mixed set of information about a medication product, Montelukast Sodium. The text includes some numerical data and some descriptions of instructions, precautions, and details for its use. It is advised for use only for pediatric patients at or above the age of 14, with a dose of 5mg per chewable tablet. The tablets come in a bottle of 90 pieces, only to be prescribed by a doctor. The text also includes information to pharmacists to hand over the patient information leaflet to each patient.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.