Product Images Fondaparinux Sodium
View Photos of Packaging, Labels & Appearance
Product Label Images
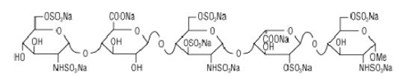
The following 13 images provide visual information about the product associated with Fondaparinux Sodium NDC 55111-678 by Dr. Reddy's Laboratories Limited, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10mg2sCarton - 10mg2sCarton

This is a description of a medication contained in a single-dose syringe. The syringe contains 10mg of fondaparinux sodium, USP, which is injected subcutaneously. The solution is isotonic and may contain pH adjusters. It is recommended to consult the package insert for dosage instructions. The syringe is affixed with an active needle protection system and contains two doses. The medication should be stored at a controlled room temperature and any unused portion should be discarded. The GTIN (bar code) for the medication is 0355111681028.*
25mg2sCarton - 25mg2sCarton

This is a description of a medication called Fondaparinux Sodium Injection. The medication comes in the form of a single-dose, prefilled syringe with 2.5 mg/0.5 mL. The solution contains fondaparinux sodium and an isotonic solution of sodium chloride and water for injection. The recommended dose is 2.5 mg for subcutaneous injection once a day. The medication should be stored at room temperature. The label on the package should be affixed to the medication.*
5mg2sCarton - 5mg2sCarton

This appears to be a description of a pharmaceutical product called "Fondapal", which is a single-dose prefilled syringe containing 5 mg of a compound called "fondaparna sodiun". The recommended dose is one injection subcutaneously per day. The product should be stored at a controlled room temperature and any unused portions should be discarded. There are also details about the product packaging and identification numbers.*
75mg2sCarton - 75mg2sCarton

This text appears to be a description of an injection drug called "Fondaparinux Sodium Injection" that comes in pre-filled syringes containing 7.5 mg/0.6 mL of the drug. The recommended dose is 0.5 mg subcutaneous injection, once daily, and it should be stored at 20-25°C (68-77°F). The injection also contains sodium chloride and water for injection and may contain pH adjusters like sodium hydroxide and/or hydrochloric acid. There are two single-dose pre-filled syringes in each container.*
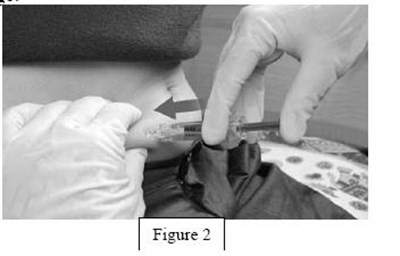
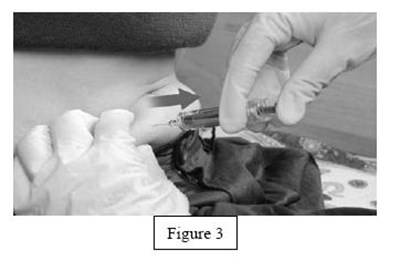
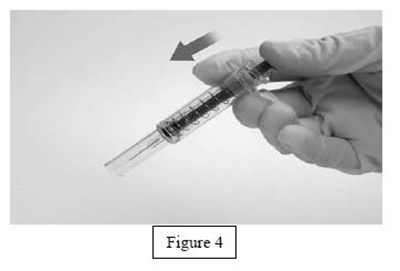
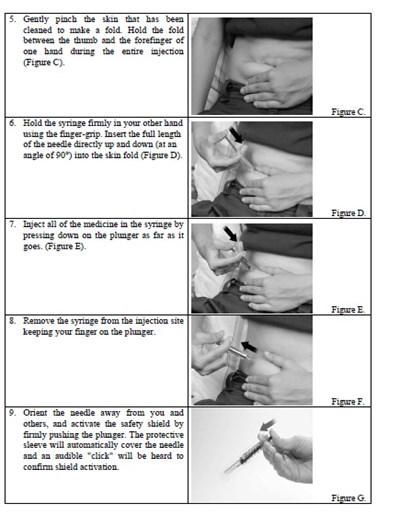
instrucn2cond - instruc2cond

This is a set of instructions for disposing of used syringes and needles containing fondaparinux sodium through a FDA-clared sharps disposal container made of a heavy-duty puncture-resistant plastic that can be sealed tightly to prevent leaks and kept stable during disposal. It is recommended to follow community guidelines for safe disposal, and for more information, consult the FDA website. It is not advised to dispose of sharps in household trash or recycling.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.