Product Images Amiodarone Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 7 images provide visual information about the product associated with Amiodarone Hydrochloride NDC 55150-182 by Auromedics Pharma Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 150 mg per 3 mL (50 mg / mL) Container Label - amiodarone fig1

AUROMEDICS' product P1416648 is an Rx-only Amiodarone Hydrochloride Injection with 150 mg per 3 mL (50 mg / mL). Each mL of injection contains 50 mg amiodarone hydrochloride USP, 100 mg polysorbate 80, and 20.2 mg benzyl alcohol in water for injection. The product must be diluted before use and is for L.V. use only. It comes in a sterile 3 mL single dose vial. It is manufactured in India for AuroMedics Pharma LLC, with a code of TS/DRUGS/13/2010. The company is based in E. Windsor, NJ 08520.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 150 mg per 3 mL (50 mg / mL) Container-Carton (10 Vials) - amiodarone fig2
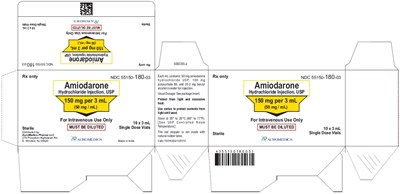
AMIODARONE HYDROCHLORIDE INJECTION, USP is a medication used for intravenous use only. The single dose vials must be diluted before use and protected from light. The usual dosage is 150 mg per 3 mL. The medication contains 50 mg of amiodarone per mL. The package insert should be consulted for more information on dosages. The medication is sterile and must be used with caution if the patient has any allergies or sensitivities to natural rubber.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 450 mg per 9 mL (50 mg / mL) Container Label - amiodarone fig3

Amiodarone Hydrochloride Injection is a sterile single dose vial for intravenous use only that must be diluted. Each vial contains 450mg per 9mL (50mg/mL) of amiodarone hydrochloride. Other ingredients include 100mg of polysorbate 80, 20.2mg of benzyl alcohol in water for injection. It is advised to see the package insert for proper dosage. The vial must be protected from light and excessive heat, and the carton should be used to protect the content of the vial. The product must be stored at a temperature between 20° to 25°C (68° to 77°F). The product is distributed by AuroMedics Pharma LLC and it is made in India.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 450 mg per 9 mL (50 mg / mL) Container-Carton (10 Vials) - amiodarone fig4

This is a product description of Amiodarone Hydrochloride Injection, USP. It is meant for intravenous use only and should be stored at a temperature between 20-25°C. The package contains 10 single-dose sterile vials and the carton should be used to protect it from light until use. It also includes the manufacturer's name, Rorticdes Parma LLC, and the distributor, AUROMEDICS.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 900 mg per 18 mL (50 mg / mL) Container Label - amiodarone fig5

This is a description for an injectable solution containing 50 mg of amiodarone hydrochloride USP, 100mg polysorbate 80 and 20.2 mg benzyl alcohol in water for injection. The recommended dosage is included in the package insert and the product should be protected from light and excessive heat. It must be stored at 20° to 25°C (68° to 77°F). It is available in a multiple dose vial and is intended for intravenous use only. This solution is distributed by AuroMedics Pharma LLC and is made in India.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 900 mg per 18 mL (50 mg / mL) Container-Carton (1 Vial) - amiodarone fig6
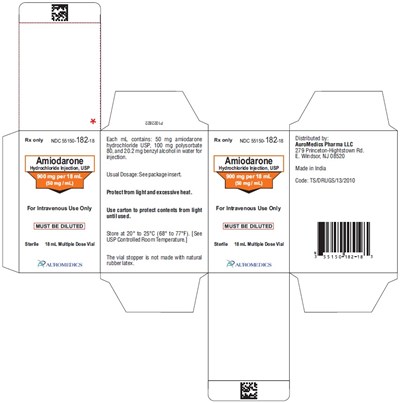
This is a description of a medication called Amiodarone. It is a Hydrochloride injection that is contained in an 18 mL multi-dose vial and must be diluted before intravenous use. Each mL of the medication contains 50 mg of Amiodarone hydrochloride USP, 100 mg Polysorbate 80, 20.2 mg Benzyl alcohol, and water for injection. The usual dosage is not specified, so it should be checked in the package insert. The contents of the vial must be protected from light until used and stored in a controlled room temperature of 20°C to 25°C (68°F to 77°F). The vial stopper is not made of natural rubber latex. It is distributed by AuroMedics Pharma LLC and made in India.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.
