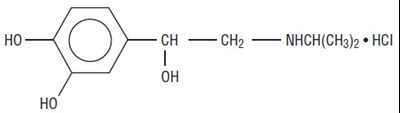
Product Images Isoproterenol Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 5 images provide visual information about the product associated with Isoproterenol Hydrochloride NDC 55150-316 by Eugia Us Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 02 mg per mL - 1 mL Ampule Label - isoproterenol fig1

This text appears to be a medication label that provides important handling and usage instructions for a drug called Isoproterenol HCI Injection. It is produced in India by AuroMedics Pharma LLC and is intended for intravenous, subcutaneous, intramuscular, or intracardiac use only. The label also indicates that the drug should be protected from light.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL – 0.2 mg per mL - 1 mL Container-Carton [25 Ampules] - isoproterenol fig2
![isoproterenol-fig2.jpg PACKAGE LABEL-PRINCIPAL DISPLAY PANEL – 0.2 mg per mL - 1 mL Container-Carton [25 Ampules] - isoproterenol fig2](https://ndclist.com/assets/spl/images/3e4050e8-7d94-4ed7-951d-1ff818ddadf1/400x-isoproterenol-fig2.jpg)
This is a description of Isoproterenol HCI Injection, USP with a concentration of 0.2 mg per mL. It is indicated for intravenous, subcutaneous, intramuscular, or intracardiac use only. Each sterile ampule contains 25x1mL of the product, with each mL consisting of 0.2 mg isoproterenol hydrochloride and other ingredients including edetate disodium, sodium chloride, sodium citrate, and citric acid. The recommended dosage is not provided. The product is sensitive to light and should be stored in an opaque container at a controlled room temperature between 20° to 25°C (68° to 77°F). The manufacturer is AuroMedics Pharma LLC, located in Windsor, NJ, and the product is made in India.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 1 mg per 5 mL (0.2 mg / mL) - 5 mL Ampule Label - isoproterenol fig3

This is a medication called Isoproterenol HCI with a dosage strength of 1mg per 5mL, with instructions that it is to be used only Intravenously, Subcutaneously, Intramuscularly or Intracardially. It should be protected from light, and its manufacturer is AuroMedics Pharma LLC. However, there seems to be some errors in the text, especially towards the end, where it is difficult to extract any meaning or continuation.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL – 1 mg per 5 mL (0.2 mg / mL) - 5 mL Container-Carton [10 Ampules] - isoproterenol fig4
![isoproterenol-fig4.jpg PACKAGE LABEL-PRINCIPAL DISPLAY PANEL – 1 mg per 5 mL (0.2 mg / mL) - 5 mL Container-Carton [10 Ampules] - isoproterenol fig4](https://ndclist.com/assets/spl/images/3e4050e8-7d94-4ed7-951d-1ff818ddadf1/400x-isoproterenol-fig4.jpg)
This is a label for Isoproterenol HCl Injection, USP which comes in 1 mg per 5 mL 10 x 5 ML ampoules for Intravenous, Subcutaneous, Intramuscular or Intracardiac Use Only. The label also includes information about the brand, dosage, storage, and distribution details.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.