Product Images Icatibant
View Photos of Packaging, Labels & Appearance
- Figure 2 Time to 50% reduction from baseline in 3-item VAS score - icatibant fig1
- Figure I - icatibant fig10
- Figure J - icatibant fig11
- Figure K - icatibant fig12
- Figure L - icatibant fig13
- Figure M - icatibant fig14
- Figure N - icatibant fig15
- Figure O - icatibant fig16
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL-30 mg per 3 mL (10 mg/mL) - Syrine Label - icatibant fig17
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL-30 mg per 3 mL (10 mg/mL) - Syringe Carton (1 Syringe) - icatibant fig18
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL-30 mg per 3 mL (10 mg/mL) - Syringe-Carton (3 Syringes) - icatibant fig19
- Figure A - icatibant fig2
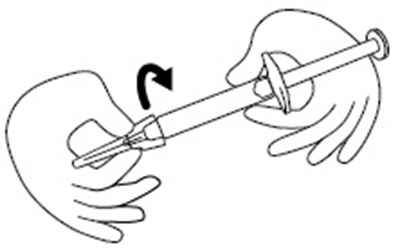
- Figure B - icatibant fig3
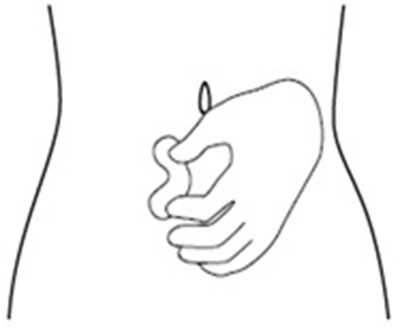
- Figure C - icatibant fig4
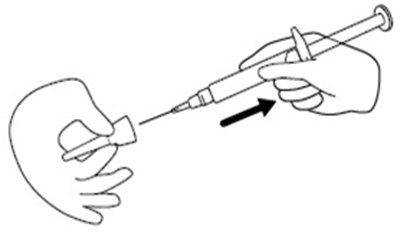
- Figure D - icatibant fig5
- Figure E - icatibant fig6
- Figure F - icatibant fig7
- Figure G - icatibant fig8
- Figure H - icatibant fig9
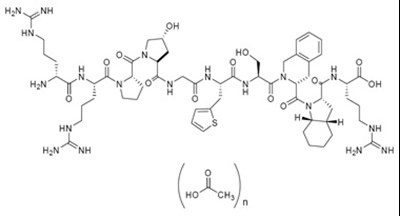
- Chemical Structure - icatibant str
Product Label Images
The following 20 images provide visual information about the product associated with Icatibant NDC 55150-351 by Eugia Us Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL-30 mg per 3 mL (10 mg/mL) - Syrine Label - icatibant fig17

Icatibant is a medication available in the form of an injection. Each injection contains 30 mg of the active ingredient per 3 mL, with a concentration of 10 mg/mL. This injection is intended for subcutaneous use only. The medication comes in a single-dose prefilled syringe with a capacity of 3 mL. It is manufactured in India for Eugia US LLC, based in E. Windsor, NJ 08520. The code displayed is TS/DRUGS/13/2010.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL-30 mg per 3 mL (10 mg/mL) - Syringe Carton (1 Syringe) - icatibant fig18

This text appears to be from the packaging of a medication called Carlon. The medication is contained in a single-dose, prefilled syringe with a protective cap. It is indicated for subcutaneous use and each syringe delivers 30 mg of icatibant. The inactive ingredients include sodium chloride, glacial acetic acid, sodium hydroxide, and water for injection. The medication should be stored between 2° to 25°C and should not be frozen. The package insert contains full prescribing information, dosage, and administration instructions. The medication does not contain preservatives and the plunger rubber stopper is not made with natural rubber latex.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL-30 mg per 3 mL (10 mg/mL) - Syringe-Carton (3 Syringes) - icatibant fig19

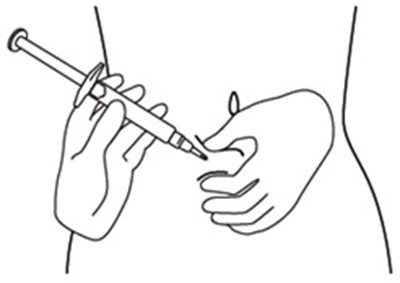
Figure A - icatibant fig2

This is a description for a pre-filled syringe with a protective cap over the syringe tip. It may come with an alcohol wipe, but the wipe is not included in the carton.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.