Product Images Methotrexate
View Photos of Packaging, Labels & Appearance
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg per 2 mL - Container Label - methotrexate fig1
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 250 mg per 10 mL - Container Label - methotrexate fig10
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 250 mg per 10 mL- Container-Carton (1's Pack) - methotrexate fig11
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg per 2 mL - Container-Carton (1's Pack) - methotrexate fig2
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg per 2 mL - Container-Carton (5's Pack) - methotrexate fig3
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg per 4 mL - Container Label - methotrexate fig4
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg per 4 mL - Container-Carton (1's Pack) - methotrexate fig5
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg per 4 mL - Container-Carton (10's Pack) - methotrexate fig6
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg per 8 mL - Container Label - methotrexate fig7
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg per 8 mL - Container-Carton (1's Pack) - methotrexate fig8
- PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg per 8 mL - Container-Carton (10's Pack) - methotrexate fig9
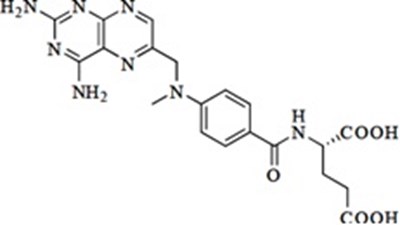
- checmical structure - methotrexate str
Product Label Images
The following 12 images provide visual information about the product associated with Methotrexate NDC 55150-513 by Eugia Us Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg per 2 mL - Container Label - methotrexate fig1

This is a prescription medication vial containing 50 mg of Methotrexate, with additional inactive ingredients such as 9.8 mg sodium chloride. The medication is intended for intravenous, intramuscular, subcutaneous, and intrathecal use only. It is advised to store the vial between 20°C to 25°C and protect it from light. The prescription is for a sterile isotonic solution manufactured by Eugia US LLC in East Windsor, NJ. The text also includes precautionary statements regarding dosage adjustments, storage conditions, and discarding unused portions as hazardous material.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 250 mg per 10 mL - Container Label - methotrexate fig10

The text provides detailed information about a medication called Methotrexate. Each vial contains 250 mg of Methotrexate, equivalent to 274.18 mg of methotrexate sodium. It includes inactive ingredients such as sodium chloride. The pH is adjusted between 7.0 to 9.0 using sodium hydroxide and/or hydrochloric acid. The medication is preservative-free and should be stored at 20-25°C (68°F to 77°F) with excursions permitted to 15-30°C (59°F to 86°F). The drug is for Intravenous, Intramuscular, Subcutaneous, and Intrathecal use only. It is a sterile isotonic solution manufactured in India. It is important to discard any unused potion. The manufacturer is Eugia US LLC based in E. Windsor, NJ 08520.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 250 mg per 10 mL- Container-Carton (1's Pack) - methotrexate fig11

This text provides information about Methotrexate Injection, a prescription-only medication used for intravenous, intramuscular, subcutaneous, and intrathecal administration. It comes in a 250 mg per 10 mL single-dose vial, with each vial containing 250 mg of Methotrexate, USP. The solution is preservative-free, sterile, and isotonic. It contains sodium chloride and may have sodium hydroxide and/or hydrochloric acid to adjust pH. The drug is considered hazardous. The storage conditions, warnings, and manufacturer details are also provided. It is intended for medical use only and should be discarded if unused.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg per 2 mL - Container-Carton (1's Pack) - methotrexate fig2

This is a description of Methotrexate Injection, a prescription medication used for intravenous, intramuscular, subcutaneous, and intrathecal administration. It comes in a single-dose vial containing 50 mg of Methotrexate in a sterile, preservative-free isotonic solution. The vial stopper is latex-free and the inactive ingredients include sodium chloride. It's important to follow the prescribing information and store the medication between 20°C to 25°C. Distributor information includes Eugia US LLC located in New Jersey, with the product being manufactured in India.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 50 mg per 2 mL - Container-Carton (5's Pack) - methotrexate fig3

This is a description of Methotrexate Injection, USP 50 mg per 2 mL, a preservative-free hazardous drug for intravenous, intramuscular, subcutaneous, and intrathecal use. It is presented in sterile isotonic solution in 5 x 2 mL single-dose vials. Each vial contains 50 mg of Methotrexate USP, equivalent to 54.8 mg of methotrexate sodium. Inactive ingredients include sodium chloride, sodium hydroxide, and hydrochloric acid to adjust pH. The medication should be stored at 20°C to 25°C (68°F to 77°F) and protected from light. The vial stoppers do not contain natural rubber latex. Distributed by Eugia US LLC in East Windsor, New Jersey, and made in India. Prescribing information is available, and caution should be taken when handling this drug.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg per 4 mL - Container Label - methotrexate fig4

This text provides details about a medication called Methotrexate, which is an injection containing 100 mg per 4 mL (25 mg/mL) in a sterile isotonic solution. The inactive ingredients include sodium chloride. The medication is stored at a temperature range of 20°C to 25°C (68°F to 77°F) with excursions permitted to 15°C to 30°C (59°F to 86°F). It is essential to protect it from light. The text also mentions a unique identifier code related to drugs and a reference to prescribing information.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg per 4 mL - Container-Carton (1's Pack) - methotrexate fig5

This is a description of Methotrexate, a prescription medication injected in a 100 mg per 4 mL (25 mg/mL) solution. It is a preservative-free hazardous drug for intravenous, intramuscular, subcutaneous, and intrathecal use only. Each single dose vial contains 100 mg of Methotrexate USP equivalent to 109.6 mg of methotrexate sodium. It also has inactive ingredients like sodium chloride. The drug should be stored between 20°C to 25°C (68°F to 77°F) with excursions permitted to 15°C to 30°C (59°F to 86°F). The vial stopper is latex-free. It is distributed by Eugia US LLC and made in India. The label provides the necessary information for safe administration and storage of Methotrexate.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 100 mg per 4 mL - Container-Carton (10's Pack) - methotrexate fig6

This is information about Methotrexate, an injectable drug available in vials containing 100 mg of Methotrexate. It is indicated for Intravenous, Intramuscular, Subcutaneous, and Intrathecal use only. The solution is preservative-free and comes in 4 mL single-dose vials. The drug is a Hazardous Drug and should be stored at 20°C to 25°C. The manufacturer is Eugia US LLC, and the product is made in India. This medication should be protected from light, and any unused portion should be discarded. For complete details, refer to the prescribing information.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg per 8 mL - Container Label - methotrexate fig7

This text describes a medication labeled as Methotrexate, which contains 200 mg per 8 mL vial. It can be used for intravenous, intramuscular, and subcutaneous administration. The medication is manufactured in India and distributed by Eugia US LLC. It is preservative-free and intended for intrathecal use only. The drug should be stored between 20°C to 25°C with excursions permitted between 15°C to 30°C and protected from light. The prescription label indicates that it is for prescription use only.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg per 8 mL - Container-Carton (1's Pack) - methotrexate fig8

This is a description of Methotrexate Injection, a prescription drug provided in a single-dose vial containing 200 mg of Methotrexate per 8 mL (25 mg/mL). It is preservative-free and considered a hazardous drug for use in intravenous, intramuscular, subcutaneous, and intrathecal applications. The sterile isotonic solution is preserved in a vial with specific storage instructions and cautionary measures. Distributed by Eugia US LLC and manufactured in India, this medication requires strict adherence to prescribing information and is critical to be used under professional medical supervision.*
PACKAGE LABEL-PRINCIPAL DISPLAY PANEL - 200 mg per 8 mL - Container-Carton (10's Pack) - methotrexate fig9

* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.