Product Images Donepezil Hydrochloride
View Photos of Packaging, Labels & Appearance
- d70042e0 d686 4ddc 9d54 c90a3f7f74b0 00
- d70042e0 d686 4ddc 9d54 c90a3f7f74b0 01
- de82e6d7 f231 449a a2aa 91e26eb50774 01
- de82e6d7 f231 449a a2aa 91e26eb50774 02
- de82e6d7 f231 449a a2aa 91e26eb50774 03
- de82e6d7 f231 449a a2aa 91e26eb50774 04
- de82e6d7 f231 449a a2aa 91e26eb50774 05
- de82e6d7 f231 449a a2aa 91e26eb50774 06
- de82e6d7 f231 449a a2aa 91e26eb50774 07
- de82e6d7 f231 449a a2aa 91e26eb50774 08
- de82e6d7 f231 449a a2aa 91e26eb50774 09
- de82e6d7 f231 449a a2aa 91e26eb50774 10
- de82e6d7 f231 449a a2aa 91e26eb50774 11
- de82e6d7 f231 449a a2aa 91e26eb50774 12
- de82e6d7 f231 449a a2aa 91e26eb50774 13
Product Label Images
The following 15 images provide visual information about the product associated with Donepezil Hydrochloride NDC 55154-7883 by Cardinal Health 107, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
d70042e0 d686 4ddc 9d54 c90a3f7f74b0 00

This is a description of a medication package for Donepezil Hydrochloride tablets. The package contains 10 tablets, with each coated tablet having 10mg of Donepezil Hydrochloride. The usual dosage and precautions are available in the product insert. The website WWW.MAJORPHARMACEUTICALS.COM is provided for information. The tablets should be stored in a tightly sealed container and away from children. The package is not child-resistant and intended for institutional use only. It should be dispensed under the supervision of a pharmacist along with a patient information sheet. This medication is manufactured by Torrent Pharmaceuticals Ltd and is distributed by Major® Pharmaceuticals through Cardinal Health.*
d70042e0 d686 4ddc 9d54 c90a3f7f74b0 01

Donepezil Hydrochloride Tablets is a medication distributed by Major Pharmaceuticals in a 10 tablet pack. Each tablet contains 5mg donepezil hydrochloride, USP. It's recommended to Store the tablets at 20° - 25° C (68 - 77° F); and it's only for Institutional Use. The tablet pack is not child-resistant, so it should be kept away from children. The medication should be dispensed in a tight, light-resistant container. There are more precise instructions and information available at www.majorpharmaceuticals.com which should be referred to before consuming the medication.*
de82e6d7 f231 449a a2aa 91e26eb50774 02
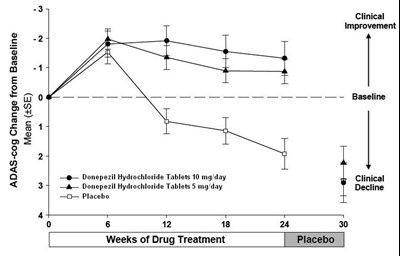
This is a table that shows the ADAS-cog change from baseline after 6, 12 and 18 weeks of drug treatment. The drugs being compared are Donepezil Hydrochloride Tablets 10 mg/day, Donepezil Hydrochloride Tablets 5 mg/day, and Placebo. The table shows mean values for clinical improvement and clinical decline.*
de82e6d7 f231 449a a2aa 91e26eb50774 03
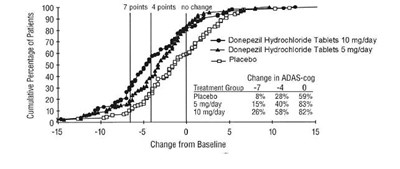
The text describes a chart showing the cumulative percentage of patients with Alzheimer's disease and the change in their cognitive function (ADAS-cog) based on the dosage of Donepezil Hydrochloride (5mg and 10mg) or a placebo over a period of 10 months. The chart also shows the percentage of patients with no change in cognitive function.*
de82e6d7 f231 449a a2aa 91e26eb50774 04
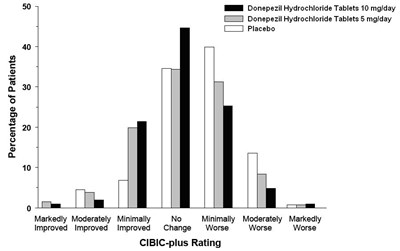
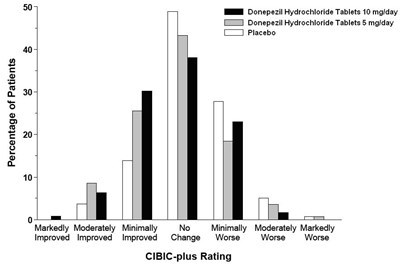
This text appears to be a chart or graph showing the effectiveness of three different treatments (BN Donepezil Hydrochloride Tablets 10 mg/day, Donepezil Hydrochloride Tablets 5 mg/day, and Placebo) for patients over a period of time. The chart appears to measure improvement, worsened symptoms, and no change using the CIBIC-plus rating system. However, without further context or a clear image, it is difficult to provide a more detailed description.*
de82e6d7 f231 449a a2aa 91e26eb50774 09
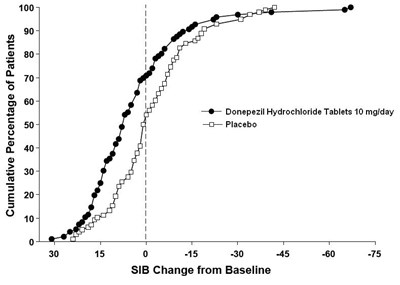
The text is describing a chart that shows the cumulative percentage of patients for Donepezil Hydrochloride Tablets 10 mg/day and placebo. It also shows the change from baseline in SIB (Severe Impairment Battery) for the patients.*
de82e6d7 f231 449a a2aa 91e26eb50774 10
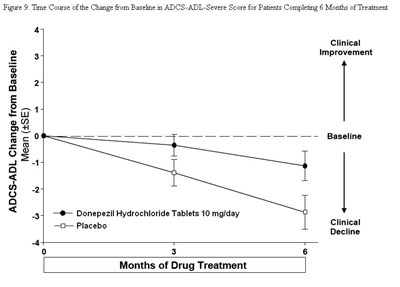
The text describes a figure showing the time course of change in a score called ADCS-ADL-Severe for patients who completed 6 months of treatment. The figure has a graph with lines indicating clinical improvement or decline in the score over time, as well as a baseline and treatment with donepezil hydrochloride tablets (10mg/day) or placebo.*
de82e6d7 f231 449a a2aa 91e26eb50774 11
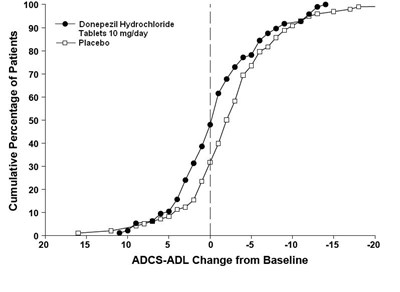
The text describes a graph showing the cumulative percentage of patients, comparing the effectiveness of Donepezil Hydrochloride (10mg/day) tablets versus a placebo, in terms of ADCS-ADL change from the baseline. The graph ranges from -10% to 100%, with the percentage values shown on the Y-axis, and the ADCS-ADL change from the baseline values on the X-axis, ranging from +5 to -5.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.