FDA Label for Diphenhydramine Hcl
View Indications, Usage & Precautions
- ACTIVE INGREDIENT(IN EACH BANDED CAPSULE)
- PURPOSE
- USES:
- WARNINGS:
- DO NOT USE
- ASK A DOCTOR OR PHARMACIST BEFORE USE
- ASK A DOCTOR OR PHARMACIST BEFORE USE OF YOU ARE
- WHEN USING THIS PRODUCT
- IF PREGNANT OR BREAST-FEEDING,
- KEEP OUT OF REACH OF CHILDREN.
- DIRECTIONS:
- OTHER INFORMATION:
- INACTIVE INGREDIENTS: BLACK IRON OXIDE, D & C RED #28, D & C BLUE #1, FD & C RED #40, GELATIN, LACTOSE MONOHYDRATE, MAGNESIUM STEARATE, SILICON DIOXIDE, SODIUM LAURYL SULFATE
- 16. HOW SUPPLIED/STORAGE AND HANDLING
- STORAGE AND HANDLING
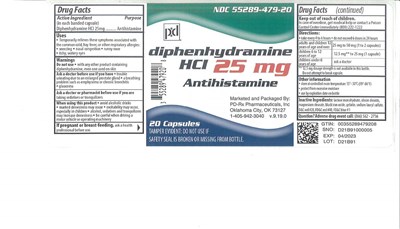
- PACKAGE LABEL.PRINCIPAL DISPLAY PANEL
Diphenhydramine Hcl Product Label
The following document was submitted to the FDA by the labeler of this product Pd-rx Pharmaceuticals, Inc.. The document includes published materials associated whith this product with the essential scientific information about this product as well as other prescribing information. Product labels may durg indications and usage, generic names, contraindications, active ingredients, strength dosage, routes of administration, appearance, warnings, inactive ingredients, etc.
Active Ingredient(In Each Banded Capsule)
Diphenhydramine HCL 25 mg
Purpose
Antihistamine
Uses:
- Temporarily relieves these symptoms due to common cold, hay fever, or other upper respiratory allergies:
-Sneezing
-watery eyes
-Runny nose
-Itchy, watery eyes
Warnings:
Do Not Use
- To make child sleepy
with any other product containing diphenhydramine, even one used on skin.
Ask A Doctor Or Pharmacist Before Use
If you have
- Trouble urinating due to an enlarged prostate gland
- A breathing problem such as emphysema or chronic bronchitis
- Glaucoma
Ask A Doctor Or Pharmacist Before Use Of You Are
taking sedatives or tranquilizers
When Using This Product
- Avoid alcoholic drinks
- Marked drowsiness may occur
- Excitability may occur, especially in children
- Alcohol, sedatives and tranquilizers may increase drowsiness
- Be careful when driving a motor vehicle or operating machinery
If Pregnant Or Breast-Feeding,
ask a health professional before use.
Keep Out Of Reach Of Children.
In case of accidental overdose, get medical help or contact a Poison Control Center immediately. (800) 222-1222.
Directions:
- Take every 4-6 hours, or as directed by a doctor
- Do not exceed 6 times in 24 hours.
| Adults and children 12 years and above
| (1 to 2 capsules)
|
| Children 6 to under 12 years
| (1 capsule)
|
| Children under 6 years
| ask a doctor
|
Other Information:
- Tamper Evident: Do not use if safety seal is broken or missing from bottle.
- Store at controlled room temperature 20°-25°C (68°-77°F)
- Protect from excessive moisture
Inactive Ingredients: Black Iron Oxide, D & C Red #28, D & C Blue #1, Fd & C Red #40, Gelatin, Lactose Monohydrate, Magnesium Stearate, Silicon Dioxide, Sodium Lauryl Sulfate
16. How Supplied/Storage And Handling
Pink and white capsule with red band, imprinted with PH014, supplied as:
Bottles of 30 (NDC 55289-479-30).
Storage And Handling
Storage
Store at room temperature 20°C to 25°C (68°F to 77°F); excursions permitted between 15°C to 30°C (59°F to 86°F) [see USP Controlled Room Temperature]
Package Label.Principal Display Panel
* Please review the disclaimer below.