Product Images Amjevita
View Photos of Packaging, Labels & Appearance
- Figure 1 - amjevita 01
- Figure 2 - amjevita 02
- Image - amjevita 03
- Image - amjevita 04
- Image - amjevita 05
- Image - amjevita 06
- Image - amjevita 07
- Image - amjevita 08
- Image - amjevita 09
- Image - amjevita 10
- Image - amjevita 11
- Image - amjevita 12
- Image - amjevita 13
- Image - amjevita 14
- Image - amjevita 15
- Image - amjevita 16
- Image - amjevita 17
- Image - amjevita 18
- Image - amjevita 19
- Image - amjevita 20
- Image - amjevita 21
- Image - amjevita 22
- Image - amjevita 23
- Image - amjevita 24
- Image - amjevita 25
- Image - amjevita 26
- Image - amjevita 27
- Image - amjevita 28
- Image - amjevita 29
- Image - amjevita 30
- Image - amjevita 31
- Image - amjevita 32
- Image - amjevita 33
- Image - amjevita 34
- Image - amjevita 35
- Image - amjevita 36
- Image - amjevita 37
- Image - amjevita 38
- Image - amjevita 39
- PRINCIPAL DISPLAY PANEL - 20 mg Syringe Carton - amjevita 40
- PRINCIPAL DISPLAY PANEL - 40 mg Syringe Carton - amjevita 41
- PRINCIPAL DISPLAY PANEL - 40 mg Autoinjector Carton - amjevita 42
Product Label Images
The following 42 images provide visual information about the product associated with Amjevita NDC 55513-480 by Amgen Inc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
Figure 1 - amjevita 01

The text appears to be a graph showing the administration of a medication (40mg every other week) compared to a placebo over a period of weeks. The label on the x-axis indicates the measurement of time.*
Figure 2 - amjevita 02

The text is describing a chart or graph showing the ASAS 20 response of a placebo group (consisting of 107 participants) compared to an adalimumab group (consisting of 208 participants) over a period of 24 weeks. The chart/plot shows time on the x-axis (measured in weeks) and the ASAS 20 response on the y-axis. Without access to the visual aids, further interpretation or insight is not feasible.*
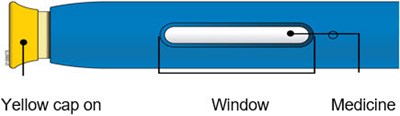
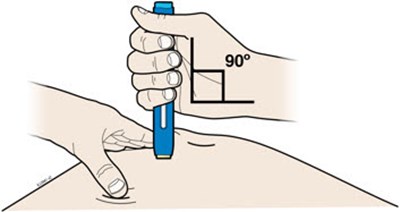
Image - amjevita 04

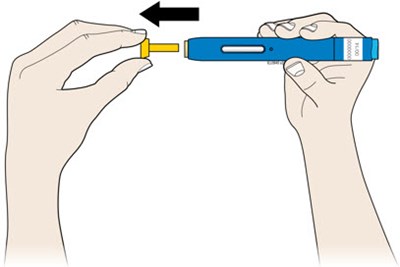
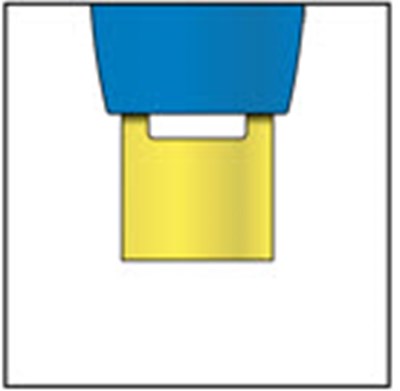
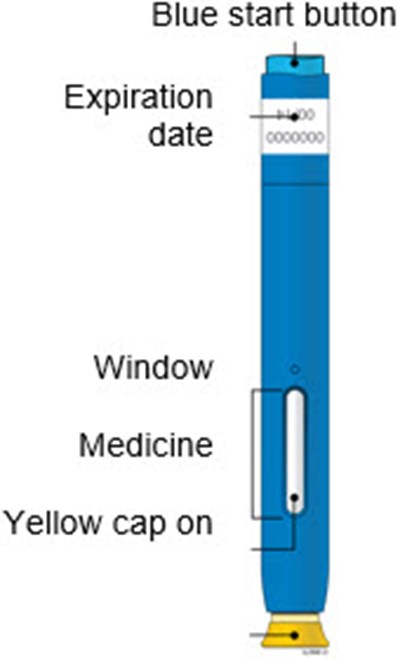
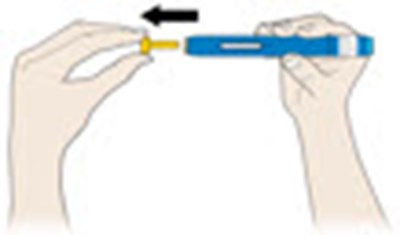
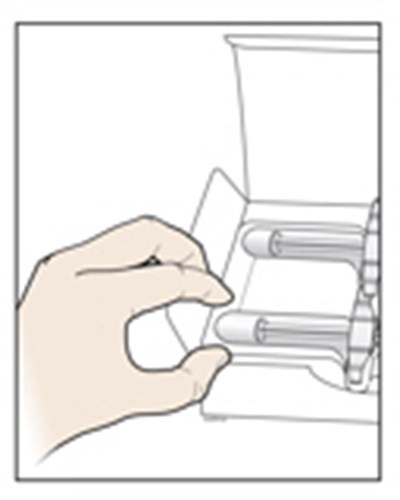
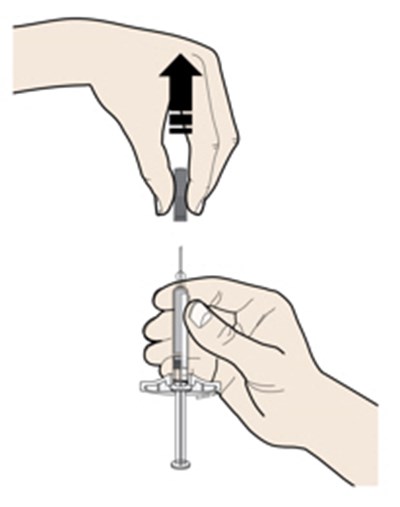
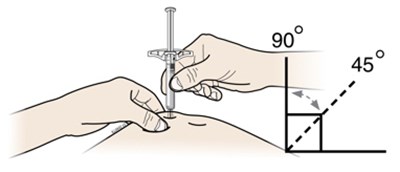
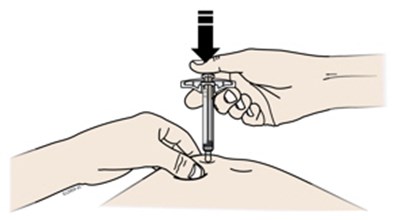
This appears to be a set of instructions or information related to a medical injection device. It includes details about the expiration date and the color-coding of various components, such as the yellow window and safety guard. It also references removing the yellow cap.*
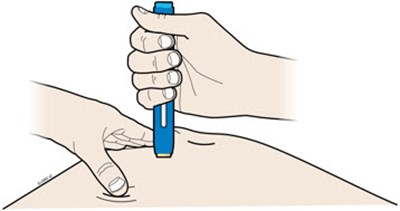
Image - amjevita 07

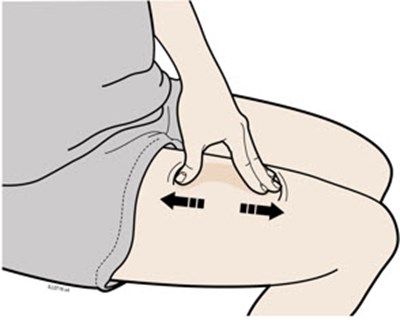
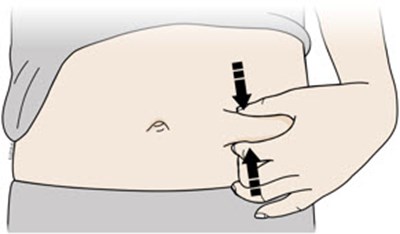
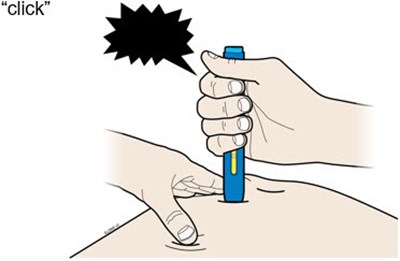
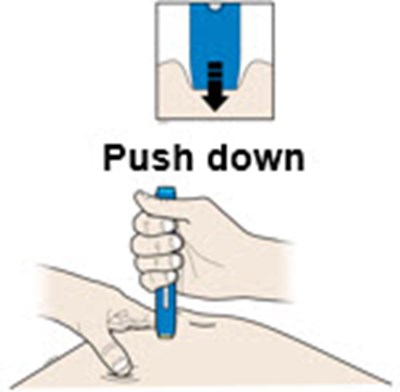
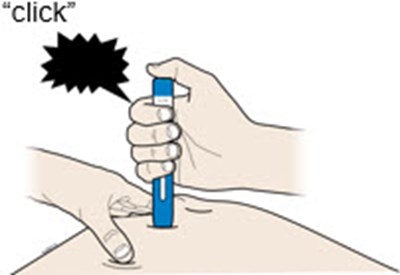
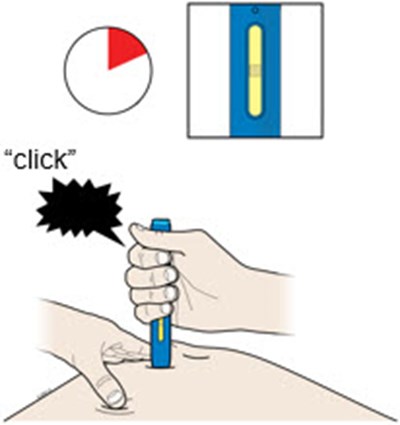
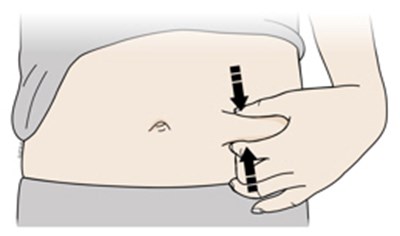
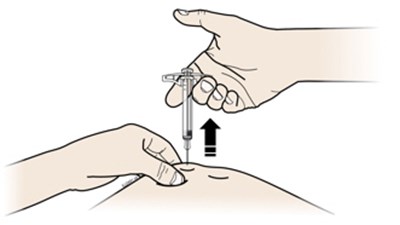
This text is describing two body parts: the stomach area (abdomen) and the thigh.*
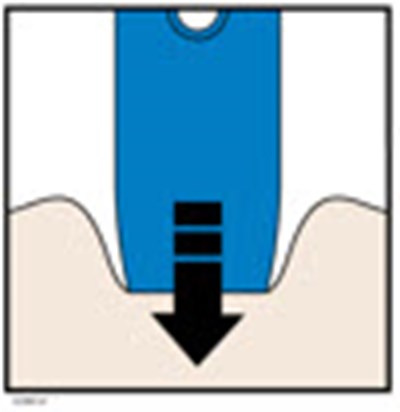
Image - amjevita 21

This appears to be a list of body parts: Stomach area (abdomen) and Thigh. No further context or description is available.*
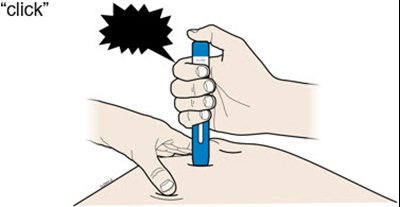
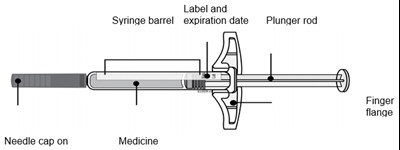
Image - amjevita 27
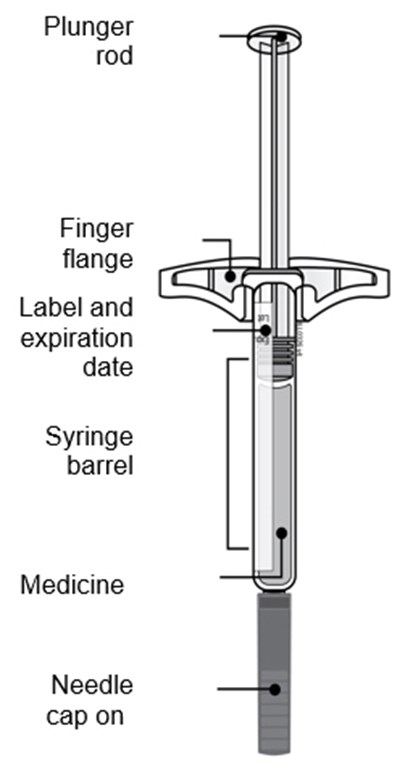
This text seems to be a list of components of a medication syringe including a plunger rod, finger flange, label with an expiration date, syringe barrel, medicine, and a needle cap.*
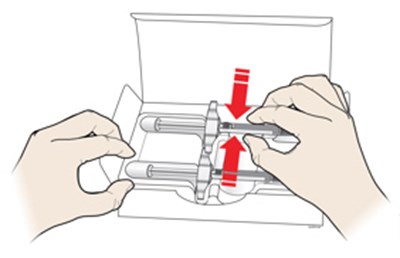
Image - amjevita 28

This appears to be a list of various components typically found in medical syringe equipment, including a used plunger rod, finger flange, label and expiration date, used syringe barrel, used needle, and needle cap. It does not provide any specific information about the condition of these items or their intended use.*
Image - amjevita 33

This is a medical term indicating two parts of the human body. The first one refers to the area of the body located between the chest and the pelvis, commonly known as the "stomach" or "abdomen". This area contains vital organs such as the stomach, liver, intestines, kidneys, and spleen. The second term refers to the upper part of the leg, extending from the hip to the knee, known as the "thigh". It contains several muscles, including the quadriceps and hamstrings, that are essential for movement and mobility.*
PRINCIPAL DISPLAY PANEL - 20 mg Syringe Carton - amjevita 40

This is a medication called Adalimumab-atto, which is an injectable solution for subcutaneous use. It comes in a single-dose prefilled syringe with a storage temperature between 2-8°C (36°F-46°F) and should not be frozen or shaken. It is intended for people who weigh between 18kg to <%0 kg and the package includes additional instructions for storage. However, the text provided is unreliable and contains errors that make the information difficult to read.*
PRINCIPAL DISPLAY PANEL - 40 mg Autoinjector Carton - amjevita 42

This appears to be a description of a medication called "fAMJEVITA" with the active ingredient "adalimumab-atto". It is a sterile solution in a prefilled autoinjector that should be stored refrigerated and protected from light, and should not be shaken or frozen. It is intended for subcutaneous use only and the enclosed medication guide is required for each use. The product is not made with natural rubber latex and should not be re-used. Other text in the description is not readable.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.