Product Images Tramadol Hydrochloride
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 6 images provide visual information about the product associated with Tramadol Hydrochloride NDC 55700-029 by Quality Care Products Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image description - New Doc 2017 12 05 (3)
.jpg)
This is a description of a drug named Tramadol HC-ER 100 mg with the manufacturer's information, a warning regarding its being habit-forming, storage instructions, an NDC number for identification, and a cautionary statement to keep the medication out of children's reach. The text advises to consult with a physician and see the manufacturer's insert for more details.*
tramadol-figure1 - tramadol figure1
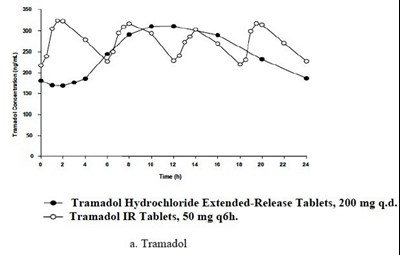
Tramadol is a medication that comes in two forms, extended-release tablets and immediate-release tablets. The extended-release tablets are taken once a day with a dose of 200 mg, while the immediate-release tablets are taken at a dose of 50 mg every six hours. This text is a consent form for tramadol treatment.*
tramadol-figure2 - tramadol figure2

Tramadol is a medication used to relieve moderate to severe pain. Tramadol Hydrochloride Extended-Release Tablets are prescribed once a day in a 200 mg dose, while Tramadol IR Tablets are prescribed to be taken four times a day in a 50 mg dose.*
tramadol-figure4 - tramadol figure4
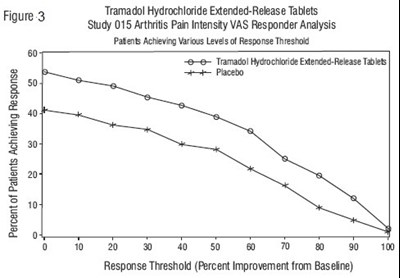
This is a description of Tramadol Hydrochloride Extended-Release Tablets, used to treat arthritis pain. The text includes a chart showing the percentage of patients achieving pain relief at various levels of response threshold.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

