Product Images Pregabalin
View Photos of Packaging, Labels & Appearance
- image description - 100mg
- image description - 150mg
- image description - 50mg
- image description - 75mg
- pregabalinchemistrystructure - pregabalin chem structure
- pregabalin-equation - pregabalin equation
- pregabalinfig1 - pregabalin fig 01
- pregabalinfig2 - pregabalin fig 02
- pregabalinfig3 - pregabalin fig 03
- pregabalinfig4 - pregabalin fig 04
- pregabalinfig5 - pregabalin fig 05
- pregabalinfig6 - pregabalin fig 06
- pregabalinfig7 - pregabalin fig 07
- pregabalinfig8 - pregabalin fig 08
- pregabalinfig9 - pregabalin fig 09
- pregabalinfig10 - pregabalin fig 10
- pregabalinfig11 - pregabalin fig 11
Product Label Images
The following 17 images provide visual information about the product associated with Pregabalin NDC 55700-774 by Quality Care Products, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image description - 100mg

Pregabalin 100 mg is a medication manufactured by Rising Pharmaceuticals and enclosed in a bottle of 30 capsules. The bottle has a GTIN number of 00355700773304 and an NDC number of 55700-0773-30. Each capsule contains Pregabalin, and the medication may be habit-forming, according to the warning label affixed to the bottle. The manufacturer, Rising Pharmaceuticals, is located in Saddle Brook, NJ, and the medication was made in Visakhapatnam, India, by Laurus Labs Limited. It is important to keep Pregabalin out of reach of children, and to store it at temperatures between 68 and 77 degrees Fahrenheit. Consultation with a physician is strongly advised, and the manufacturer's insert should be referred to for additional information. The product is contained in a red capsule marked LA;44.*
image description - 150mg
This is a description of a medication named Pregabalin 150mg in capsule form manufactured by Laurus Labs Limited, and distributed by W Care Products. The package contains 30 capsules with a white body and LA;45 marking. The medication may be habit-forming, and it is advised to consult a physician before use. The package includes a manufacturer's insert with further instructions. It is available in 1-800-337-8603 for queries. However, it is necessary to keep the medication out of reach of children and it should be stored at a temperature between 68 to 77 degrees Fahrenheit. The other details like serial number, lot number, expiration date and manufacturing and packaging details are mentioned as well.*
image description - 50mg

This is a warning label for a medication called Pregabalin 50mg, which is in capsule form. The medication should be stored at temperatures between 68 and 77 degrees Fahrenheit and kept out of reach from children. It is advised to consult with a physician and refer to the manufacturer's insert. The label cautions that the medication may be habit-forming. The capsules are manufactured by Laurus Labs Limited in India and distributed by Rising Pharmaceuticals in New Jersey.*
image description - 75mg

This text is a warning label for Pregabalin 75 mg capsules, a medication that may be habit-forming. The medication is packaged and distributed by MW @cue p woducts and is available in packages of 30 capsules. Each capsule contains Pregabalin and comes with a lot number and an expiration date. The medication is manufactured by Laurus Labs Limited in Visakhapatnam, India and distributed by Rising Pharmaceuticals, Inc. in Saddle Brook, NJ. The full prescribing information for the medication is available, and it should be stored at 68 to 77 degrees Fahrenheit. This product should be kept out of reach of children.*
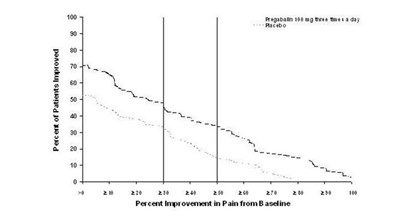
pregabalinfig3 - pregabalin fig 03

The text is not well formatted and appears to contain some kind of medical statistics, dosages, and medication names. It is not possible to generate a useful description without further context or clarification.*
pregabalinfig5 - pregabalin fig 05
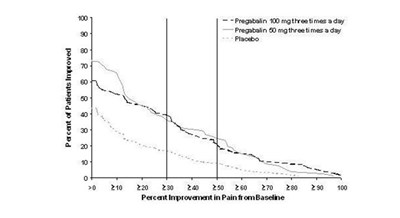
This appears to be a chart comparing the effectiveness of Fregabalin, Pregataiin, and a placebo in improving pain for patients. The dosage for Fregabalin is 100mg three times a day, while the dosage for Pregataiin is 80mg three times a day. The chart shows the percentage of patients improved and the percentage improvement in pain from the baseline for each treatment.*
pregabalinfig8 - pregabalin fig 08

This appears to be a chart containing medication dosage information and percentage improvement in pain from baseline for a particular medication called Pregabalin. It is not possible to provide additional information without further context or a clearer image of the chart.*
pregabalinfig9 - pregabalin fig 09

The text is not clear enough to generate a useful description. Please provide a readable text for evaluation.*
pregabalinfig11 - pregabalin fig 11

This appears to be a partial data chart displaying the percent improvement of subjects in a study from baseline to week 16 using pregabalin and a placebo. The chart shows numbers ranging from 100 to 280 on the y-axis and 210 to 270 on the x-axis. The percentage of subjects improved is listed on the y-axis while the drugs being used are listed on the x-axis. However, without further context or explanation, the actual purpose of the study or the full extent of the data cannot be determined with certainty.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.