Product Images Buprenorphine Hydrochloride And Naloxone Hydrochloride Dihydrate
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 4 images provide visual information about the product associated with Buprenorphine Hydrochloride And Naloxone Hydrochloride Dihydrate NDC 55700-901 by Quality Care Products, Llc, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
image description - CamScanner 03 04 2021 09.00 1

This is a warning label for a drug that contains Buprenorphine and Naloxone with strength of 2mg/0.5mg. The packaging has 30 sublingual tablets and it is produced by SpecGx LLC from Webster Groves in the USA. The label warns that the drug may be habit-forming and should be kept out of the reach of children. The lot number and expiry date were not captured in the text provided.*
Buprenorphine Chemical Structure - buprenorphine hydrochloride and naloxone hydrochlo 1
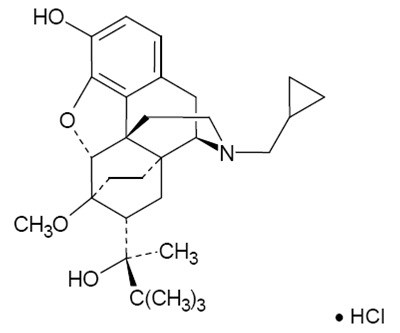
HCI stands for "Human-Computer Interaction". It is the study of how people interact with computer systems and the design of interfaces that effectively support user needs and goals. This field encompasses various aspects such as user interface design, usability testing, user research, information architecture, and cognitive psychology. The goal is to create technology that is user-friendly, efficient, and enjoyable to use.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.

