Product Images Amlodipine Besylate And Benazepril Hydrochloride
View Photos of Packaging, Labels & Appearance
- Package Label – 10 mg/ 20 mg - AmlBen10.20Lab00
- 10 mg/ 20 mg - AmlBen10.20Lab08
- 10 mg/ 20 mg - AmlBen10.20Lab09
- 10 mg/ 20 mg - AmlBen10.20Lab30
- Package Label – 10 mg/ 40 mg - AmlBen10.40Lab00
- 10 mg/ 40 mg - AmlBen10.40Lab08
- 10 mg/ 40 mg - AmlBen10.40Lab09
- 10 mg/ 40 mg - AmlBen10.40Lab30
- Package Label – 2.5 mg/ 10 mg - AmlBen2.5.10Lab00
- 2.5 mg/ 10 mg - AmlBen2.5.10Lab08
- 2.5 mg/ 10 mg - AmlBen2.5.10Lab09
- 2.5 mg/ 10 mg - AmlBen2.5.10Lab30
- Package Label – 5 mg/ 10 mg - AmlBen5.10Lab00
- 5 mg/ 10 mg - AmlBen5.10Lab08
- 5 mg/ 10 mg - AmlBen5.10Lab09
- 5 mg/ 10 mg - AmlBen5.10Lab30
- Package Label – 5 mg/ 20 mg - AmlBen5.20Lab00
- 5 mg/ 20 mg - AmlBen5.20Lab08
- 5 mg/ 20 mg - AmlBen5.20Lab09
- 5 mg/ 20 mg - AmlBen5.20Lab30
- Package Label – 5 mg/ 40 mg - AmlBen5.40Lab00
- 5 mg/ 40 mg - AmlBen5.40Lab08
- 5 mg/ 40 mg - AmlBen5.40Lab09
- 5 mg/ 40 mg - AmlBen5.40Lab30
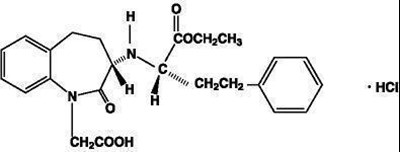
- Benazepril hydrochloride structural formula - AmlBenChemStruc1
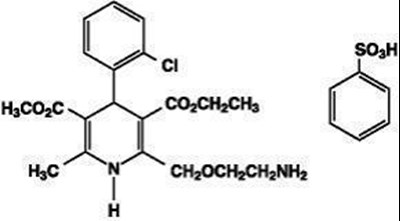
- Amlodipine besylate structural formula - AmlBenChemStruc2
Product Label Images
The following 26 images provide visual information about the product associated with Amlodipine Besylate And Benazepril Hydrochloride NDC 58118-2271 by Clinical Solutions Wholesale, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
10 mg/ 20 mg - AmlBen10.20Lab08

This text provides information about a medication called Amlodipine Besylate and Benazepril Tablets containing 10-20 MG strength. It is supplied in a pack of 30 capsules with NDC code 58118-2274-8. The prescription was filled on 03/13/13 with lot number 12310thum manufactured by Sandoz Inc. It is distributed by Clinical Solutions, a wholesaler based in Nashville, TN.*
10 mg/ 20 mg - AmlBen10.20Lab09

This text provides information about a medication package, including the NDC number, the medication name (Amlodipine Besylate and Benazepril HCL Tablets), strength (10-20 MG), quantity (30 capsules), fill date (03/13/13), lot number, expiration date (03/15/15), and the manufacturer (Sandoz Inc.). It is a prescription-only medication used for clinical solutions.*
10 mg/ 20 mg - AmlBen10.20Lab30

This text provides information about a medication with the NDC code 58118-2274-3. It is Amlodipine Besylate and Enalapril Tablets with a strength of 10-20 MG in a package containing 30 capsules. The medication was filled on 03/13/13, and the lot number is 12310thum. The manufacturer's NDC is 0781-2274-01, with an expiration date of 03/15. The manufacturer is Sandoz Inc. Additionally, there is a reference to "CLINICAL solutions."*
Package Label – 10 mg/ 40 mg - AmlBen10.40Lab00

This is a description for a Blue/Capsule form of Lotrel medication manufactured by Sandoz. The lot number is 0379 and the expiration date is 5811822790. The contact information for Clinical Solutions is provided as 877-826-5488.*
10 mg/ 40 mg - AmlBen10.40Lab08

This text contains information on a clinical solution identified as Amlodipine Besylate and Benazepril HCL tablets. The prescription was filled on 03/13/13, and the lot number is 1230 with an expiration date of 03/MSM5. The manufacturer is Sandoz Inc. The NDC number for the product is 0781-2278-01. It comes in a strength of 10-40 MG in a pack of 30 capsules, available through prescription only.*
10 mg/ 40 mg - AmlBen10.40Lab09

This text provides information about a medication called Amlodipine Besylate and Benazepril Hydrochloride Tablets in 10/40 mg strength. It includes details such as the NDC number, quantity of capsules (90), fill date, expiration date, manufacturer (Sandoz Inc.), and RX only designation.*
10 mg/ 40 mg - AmlBen10.40Lab30

This text provides information about a clinical solution product with the NDC code 58118-2279-3, containing Amlodipine Besylate and Benazepril Hydrochloride Tablets in doses of 10-40 MG. It is available in a pack of 30 capsules, and the lot number is 123lothum. The expiration date is 03/15 and it is manufactured by Sandoz Inc. The product is distributed by GL Solutions Wholesale in Nashville, TN.*
Package Label – 2.5 mg/ 10 mg - AmlBen2.5.10Lab00

This appears to be a prescription label for a medication called Amlodipine-Benazepril. It contains 2.5-10mg of the medication in white and yellow capsule form. The manufacturer is Sandoz with lot exp date 5811822710. For further details or inquiries, contact Clinical Solutions at 877-826-5488.*
2.5 mg/ 10 mg - AmlBen2.5.10Lab30

This is a medication called Amlodipine Besylate and Benazepril tablets in 25-10mg strength, containing 30 capsules. It was filled on March 13, 2013, with NDC number 58118-2271-3 by the manufacturer Sandoz Inc.*
5 mg/ 10 mg - AmlBen5.10Lab08

This text includes information about Amlodipine Besylate tablets with a concentration of 5mg, available in a bottle containing 30 capsules. The prescription drug is to be used under medical supervision only. The manufacturer and expiration date are mentioned. The product seems to be distributed by Clinical Solutions based in White's Creek, Tennessee.*
5 mg/ 10 mg - AmlBen5.10Lab09

This is a clinical solution prescription for Amlodipine Besylate and Enalapril tablets (5mg). The prescription includes 90 capsules and was filled on 03/13/13 with an expiration date of 03/15/15. The manufacturer is Sandoz Inc.*
5 mg/ 10 mg - AmlBen5.10Lab30

This is a description of pharmaceutical items distributed by Cirkal Solutions in Nashville, TN. The product is Amlodipine Besylate and Benazepril HCL Tablets USP in 5-10 mg strength. The prescription is filled with 30 capsules and was filled on 03/13/13. The manufacturer is Sandoz Inc.*
5 mg/ 20 mg - AmlBen5.20Lab08

This is a medication package containing Amlodipine Besylate and Benazepril HCL Tablets, each tablet containing 5/20mg. The package includes 30 capsules. The medication was filled on 03/13/13 and has an expiration date of 03/15. It is manufactured by Sandoz Inc.*
5 mg/ 20 mg - AmlBen5.20Lab09

This is a clinical solution in the form of Amlodipine Besylate and Benazepril HCL Tablets. The prescription is for 5-20mg tablets, with 90 capsules in each container. The lot number is 12310, manufactured by Sandoz Inc. The medication was filled on 03/13/13 and has an expiration date of 03/15/15. The National Drug Code (NDC) for this product is 58118-2273-9. This is a prescription-only medication.*
5 mg/ 20 mg - AmlBen5.20Lab30

This is the information for a prescription of Amlodipine Besylate and Enalapril tablets, 5-20mg, containing 30 capsules. The prescription was filled on 03/13/13 and the lot number is 123lothum. The manufacturer is Sandoz Inc.*
Package Label – 5 mg/ 40 mg - AmlBen5.40Lab00

This is a description of a medicine called Amlodipine-Benazepril with a dosage of 5-40mg in blue capsule form. The manufacturer is Sandoz and the lot number is 0348. For more details, contact Clinical Solutions at 877-826-5488.*
5 mg/ 40 mg - AmlBen5.40Lab08

This text provides details about a medication called Amlodipine Besylate and Benazepril Tablets in the strength of 5-40mg. The National Drug Code (NDC) for this product is 58118-2277-8, and it is available in the form of 30 capsules. The prescription was filled on 03/13/13. The expiration date is 03/15/15. The manufacturer is Sandoz Inc. These capsules were distributed by a company called Doherty Clinical Solutions located in Nashville, TN.*
5 mg/ 40 mg - AmlBen5.40Lab09

This text provides information about a pharmaceutical product, the Amlodipine Besylate and Enalapril HC tablets. The NDC code for the product is 58118-2277-9. The tablets are in 5/20mg strength and come in a package with 90 capsules. The manufacturer is Sandoz Inc. and the lot number is 1230otnum. The product expiry date is March 15, 2015. This medication is available by prescription only.*
5 mg/ 40 mg - AmlBen5.40Lab30

This text provides information about a medication called Amlodipine Besylate and Benazepril HCL Tablets, with a strength of 5/40mg. It comes in a package of 30 capsules with an NDC number of 58118-2277-3. The prescription was filled on 03/13/13 with lot number 12310thum and an expiry date of 03/15. The manufacturer is Sandoz Inc.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.