Product Images Shingrix
View Photos of Packaging, Labels & Appearance
Product Label Images
The following 10 images provide visual information about the product associated with Shingrix NDC 58160-823 by Glaxosmithkline Biologicals Sa, such as packaging, labeling, and the appearance of the drug itself. This resource could be helpful for medical professionals, pharmacists, and patients seeking to verify medication information and ensure they have the correct product.
shingrix-spl-graphic-01 - shingrix spl graphic 01
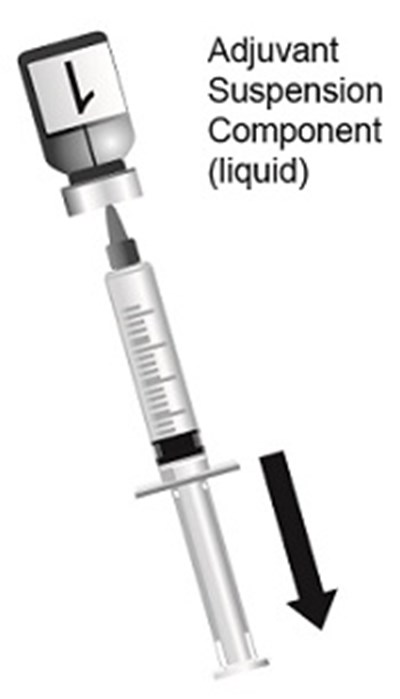
This text provides a list of words - Adjuvant, Suspension, and Component - followed by the description "liquid". It appears to be a partial list of components for a product or formula.*
shingrix-spl-graphic-02 - shingrix spl graphic 02
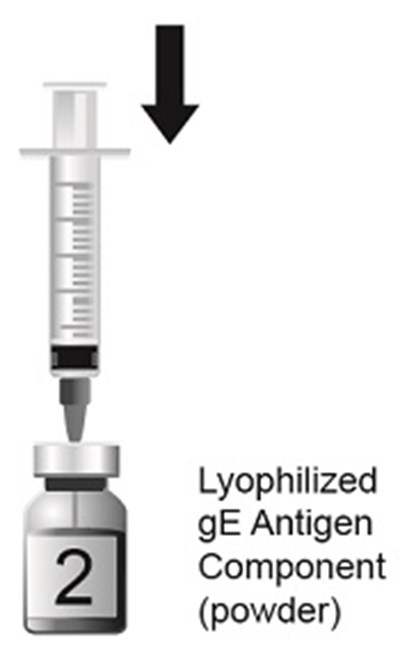
This is a description of a lyophilized (freeze-dried) powder form of an antigen component called gE. No further information is provided.*
Shingrix 1 count carton - shingrix spl graphic 05

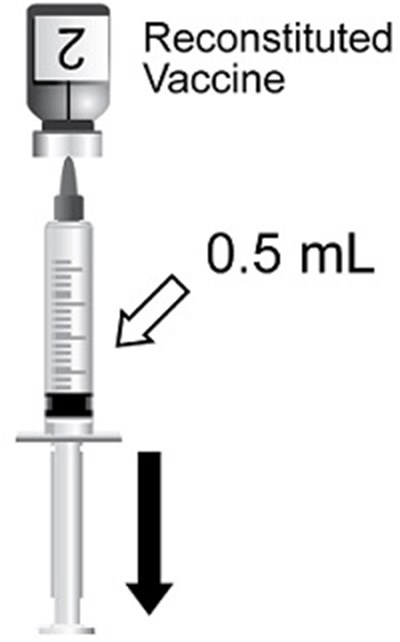
This text is a description of a vaccine called SHINGRIX with NDC number 58160-819-12. It contains a lyophilized powder and a liquid suspension which should be combined before use. The vaccine is for individuals who are 50 years or older and those who are immunocompromised and at an increased risk of herpes zoster. The vaccine contains a g lyophilized gE antigen component and a g adjuvant suspension component with a single dose of 0.5 mL after reconstitution.*
Shingrix 1 count Adjuvant vial label - shingrix spl graphic 06

This appears to be a product label for a pharmaceutical product called SHINGRIX H. The label contains important information for medical practitioners or patients about the product's components, administration, and advice on its use. The product is manufactured by GlaxoSmithKline Biologicals.*
Shingirx 1 count Antigen vial label - shingrix spl graphic 07

This is a pharmaceutical product for SHINGRIX E. It contains lyophilized gE antigen component and should not be used alone. It needs to be reconstituted with the adjuvant suspension component to form a single dose of 0.5mL after reconstitution. The manufacturer is GlaxoSmithKline Biologicals. The lot number is SEBI6Y and the expiration date is not available. The product has an NDC code of 58160-828-01.*
Shingrix 10 count carton - shingrix spl graphic 08

This is a prescription drug with NDC number 58160-823-11. It is a recombinant, adjuvanted Zoster vaccine named SHINGRIX. The drug is supplied as one vial of lyophilized powder and one vial of liquid suspension, which must be combined before use to produce a single 0.5 mL dose. It is indicated for individuals of 50 years of age or older who are immunocompromised and at high risk of Herpes Zoster. The package contains ten doses of SHINGRIX.*
Shingrix 10 count adjuvant vial label - shingrix spl graphic 09

This is a product label for one of the components required to create the SHINGRIX vaccine. It includes information on dosage, usage and manufacturer details.*
Shingrix 10 count antigen vial label - shingrix spl graphic 10

This is a product description for a vaccine called SHINGRIX. It has an NDC number of 58160-828-03, and comes in a two-part vial. The first vial contains lyophilized gE antigen component, which should not be used alone. The second vial contains an adjuvant suspension component to be used to reconstitute the antigen component. After reconstitution, the product creates a 0.5mL single-dose vaccine. It is manufactured by GlaxoSmithKline Biologicals. The remaining text is not readable.*
* The product label images have been analyzed using a combination of traditional computing and machine learning techniques. It should be noted that the descriptions provided may not be entirely accurate as they are experimental in nature. Use the information in this page at your own discretion and risk.